Draw the structure of B.
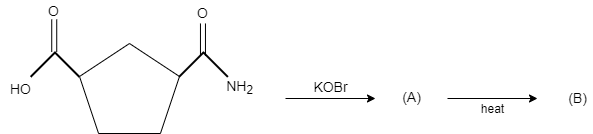
A.

B.
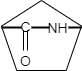
C.
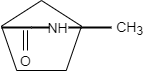
D.






Answer
574.2k+ views
Hint: In Hoffmann bromamide reaction, amide is reacted with bromine and aqueous sodium hydroxide, that leads to the formation of primary amine. In this reaction, degradation of amide takes place. The primary amine that is formed as a product contains one less carbon atom than the number of carbon atoms present in the amide.
Complete step by step answer:
Hoffmann bromamide reaction is a reaction in which a strong base is used as an alkali to attack amides, which leads to deprotonation. This reaction converts a primary amide into a primary amine and removes one carbon atom.
In this question, bromine is reacted with potassium hydroxide which leads to the formation of potassium hypobromite $(KOBr)$, that transfers the primary amide into an isocyanate intermediate. After that, isocyanate is attacked by water which leads to deprotonation.
The heat is used for the removal of carbon dioxide and quenching of ammonium cation to the required amine product.

So, the correct answer is Option B.
Additional information:
Let us now discuss the mechanism of Hoffmann bromamide reaction.
Step 1 – Strong base that contains hydroxide ion attacks the amide. Now, deprotonation of amide takes place, that leads to the formation of water and the anion of amide.
Step 2 – Now, the diatomic bromine is attacked by an anion in an alpha substitution reaction. The bromine – bromine bond breaks and this leads to the formation of $N-$ bromamide.
Step 3 – Now, the $N-$ bromamide is attacked by the base which leads to proton transfer and the formation of water with bromamide anion.
Step 4 – Now, rearrangement of bromamide takes place where the carbon group is bonded with carbonyl carbon, now it is bonded with nitrogen. This leads to the formation of isocyanate.
Step 5 – Carbamic acid is formed due to the addition of water to isocyanate.
Step 6 – The carbamic acid loses carbon dioxide and gives a negative charge on nitrogen, that is bonded to one hydrogen and the other carbon. When this is protonated by water, formation of primary amine takes place.
Note: In hoffmann bromamide reaction, The formation of an intermediate nitrene is not possible because it also implies the formation of a hydroxamic acid as a byproduct, which has never been observed. The intermediate isocyanate is hydrolyzed to a primary amine, giving off carbon dioxide.
Complete step by step answer:
Hoffmann bromamide reaction is a reaction in which a strong base is used as an alkali to attack amides, which leads to deprotonation. This reaction converts a primary amide into a primary amine and removes one carbon atom.
In this question, bromine is reacted with potassium hydroxide which leads to the formation of potassium hypobromite $(KOBr)$, that transfers the primary amide into an isocyanate intermediate. After that, isocyanate is attacked by water which leads to deprotonation.
The heat is used for the removal of carbon dioxide and quenching of ammonium cation to the required amine product.

So, the correct answer is Option B.
Additional information:
Let us now discuss the mechanism of Hoffmann bromamide reaction.
Step 1 – Strong base that contains hydroxide ion attacks the amide. Now, deprotonation of amide takes place, that leads to the formation of water and the anion of amide.
Step 2 – Now, the diatomic bromine is attacked by an anion in an alpha substitution reaction. The bromine – bromine bond breaks and this leads to the formation of $N-$ bromamide.
Step 3 – Now, the $N-$ bromamide is attacked by the base which leads to proton transfer and the formation of water with bromamide anion.
Step 4 – Now, rearrangement of bromamide takes place where the carbon group is bonded with carbonyl carbon, now it is bonded with nitrogen. This leads to the formation of isocyanate.
Step 5 – Carbamic acid is formed due to the addition of water to isocyanate.
Step 6 – The carbamic acid loses carbon dioxide and gives a negative charge on nitrogen, that is bonded to one hydrogen and the other carbon. When this is protonated by water, formation of primary amine takes place.
Note: In hoffmann bromamide reaction, The formation of an intermediate nitrene is not possible because it also implies the formation of a hydroxamic acid as a byproduct, which has never been observed. The intermediate isocyanate is hydrolyzed to a primary amine, giving off carbon dioxide.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




