Dacron is a polymer of:
A. Ethylene glycol and formaldehyde
B. Ethylene glycol and phenol
C. Ethylene glycol and phthalic acid
D. Ethylene glycol and terephthalic acid
Answer
624.9k+ views
Hint: Think about the common name of dacron; terylene. It is used regularly in the production of various fabrics. The name will give you an idea about the unit molecules involved in the monomeric unit.
Complete answer:
We know that the common name of dacron is terylene. The name is derived from one of the molecules involved in the making of its monomer. Ethylene glycol and terephthalic acid undergo condensation for the monomer of dacron. Following is the reaction that terephthalic acid and ethylene glycol undergo to form the monomer unit of dacron:
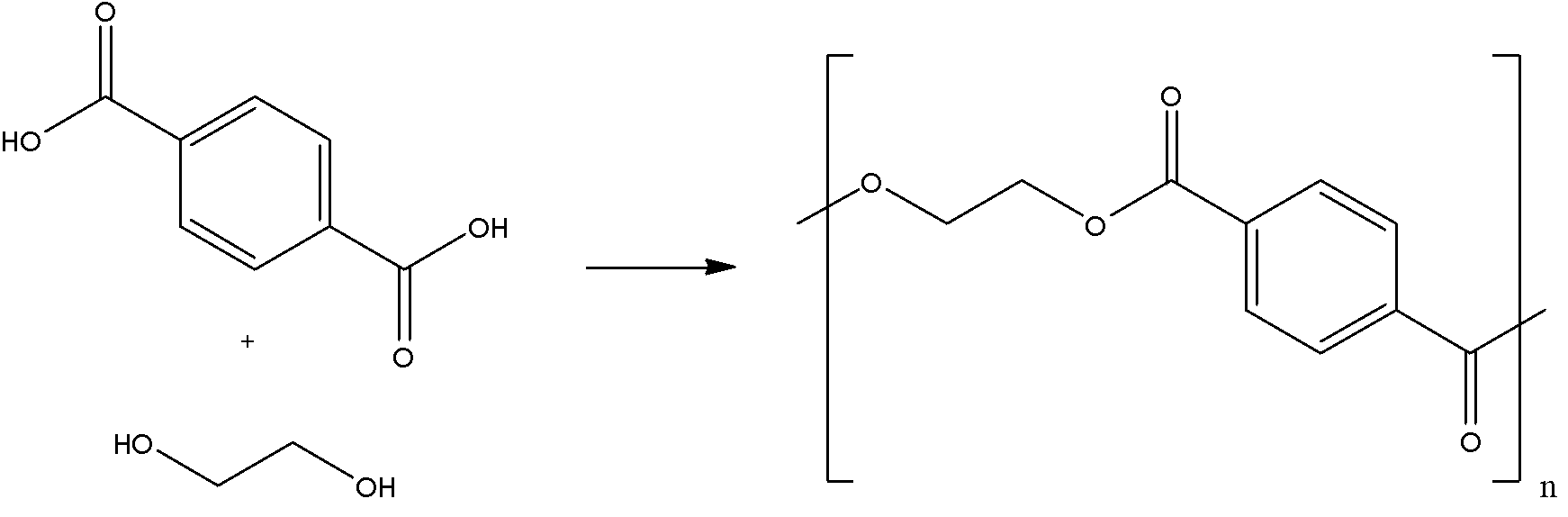
Here, we can see that one ${{H}_{2}}O$ molecule is lost to form the unit. Thus, it is called a condensation reaction.
So, the correct answer is “Option D”.
Additional Information: Dacron is a polymer that has a fairly widespread use in fabrics. It has high tensile strength and is fairly resistant to stretching whether the fabric is wet or dry. It has a high resistance to being degraded by chemical bleaches and abrasive materials. Various fabrics in which it is used include curtains, firefighter hoses, formal shirts and mixing with other woolen wear.
Note:Please do not get confused between phthalic acid and terephthalic acid. They have the same molecular formula but are structural isomers of each other. Both have two acid groups attached to a benzene ring. The difference is that while terephthalic acid is a para-isomer, phthalic acid is the ortho-isomer of the same compound.
Complete answer:
We know that the common name of dacron is terylene. The name is derived from one of the molecules involved in the making of its monomer. Ethylene glycol and terephthalic acid undergo condensation for the monomer of dacron. Following is the reaction that terephthalic acid and ethylene glycol undergo to form the monomer unit of dacron:

Here, we can see that one ${{H}_{2}}O$ molecule is lost to form the unit. Thus, it is called a condensation reaction.
So, the correct answer is “Option D”.
Additional Information: Dacron is a polymer that has a fairly widespread use in fabrics. It has high tensile strength and is fairly resistant to stretching whether the fabric is wet or dry. It has a high resistance to being degraded by chemical bleaches and abrasive materials. Various fabrics in which it is used include curtains, firefighter hoses, formal shirts and mixing with other woolen wear.
Note:Please do not get confused between phthalic acid and terephthalic acid. They have the same molecular formula but are structural isomers of each other. Both have two acid groups attached to a benzene ring. The difference is that while terephthalic acid is a para-isomer, phthalic acid is the ortho-isomer of the same compound.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




