How will you convert benzonitrile to Benzophenone?
Answer
626.4k+ views
Hint:
Benzonitrile is the chemical compound which is a colorless liquid with a sweet almond odor whereas benzophenone is a white solid that is soluble in organic solvents.
Complete step by step answer:
-Conversions are described as ratios of how much of a reactant has reacted, how much of a desired product was formed in ratio to the undesired product.
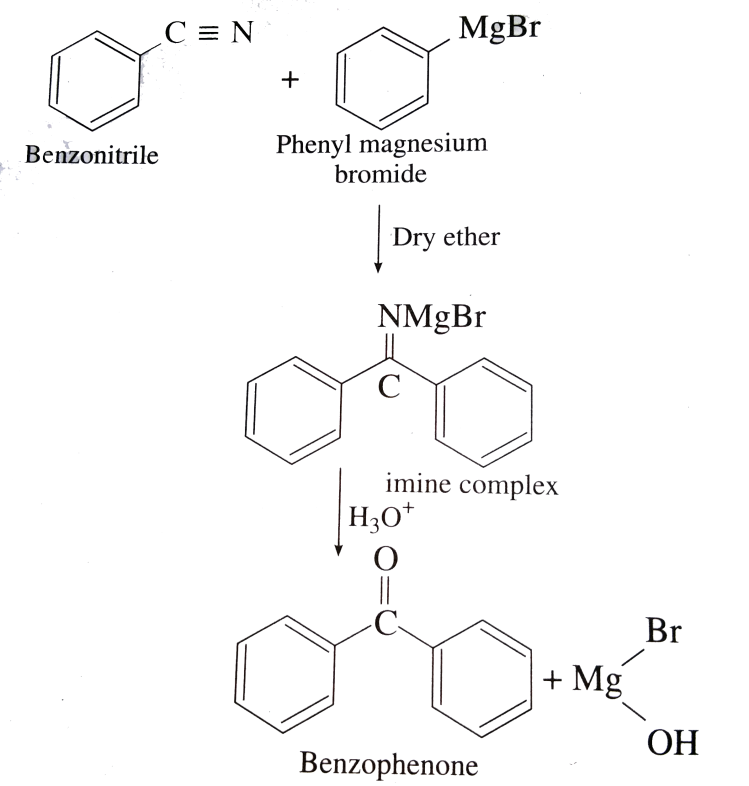
-Benzonitrile reacts with phenyl magnesium bromide in presence of dry ether to give an imine complex which on acid hydrolysis gives benzophenone.
-During the reaction benzonitrile and phenyl magnesium bromide should be taken in equimolecular proportion.
Benzophenone can also be prepared by Friedel-Crafts acylation of benzene with benzoyl chloride in the presence of a Lewis acid. (Example aluminium chloride) catalyst.
Note:
Benzophenone is used as an ultraviolet curing agent, flavor ingredient, fragrance enhancer and perfume fixate, and as an additive for plastics, coatings and adhesive formulations. Further, benzonitrile is used as a speciality solvent and to make other chemicals.
Benzonitrile is the chemical compound which is a colorless liquid with a sweet almond odor whereas benzophenone is a white solid that is soluble in organic solvents.
Complete step by step answer:
-Conversions are described as ratios of how much of a reactant has reacted, how much of a desired product was formed in ratio to the undesired product.
-Benzonitrile reacts with phenyl magnesium bromide in presence of dry ether to give an imine complex which on acid hydrolysis gives benzophenone.
-During the reaction benzonitrile and phenyl magnesium bromide should be taken in equimolecular proportion.

Benzophenone can also be prepared by Friedel-Crafts acylation of benzene with benzoyl chloride in the presence of a Lewis acid. (Example aluminium chloride) catalyst.
Note:
Benzophenone is used as an ultraviolet curing agent, flavor ingredient, fragrance enhancer and perfume fixate, and as an additive for plastics, coatings and adhesive formulations. Further, benzonitrile is used as a speciality solvent and to make other chemicals.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




