Consider the decay of a free neutron at rest: $n \to p + {e^ - }$ .
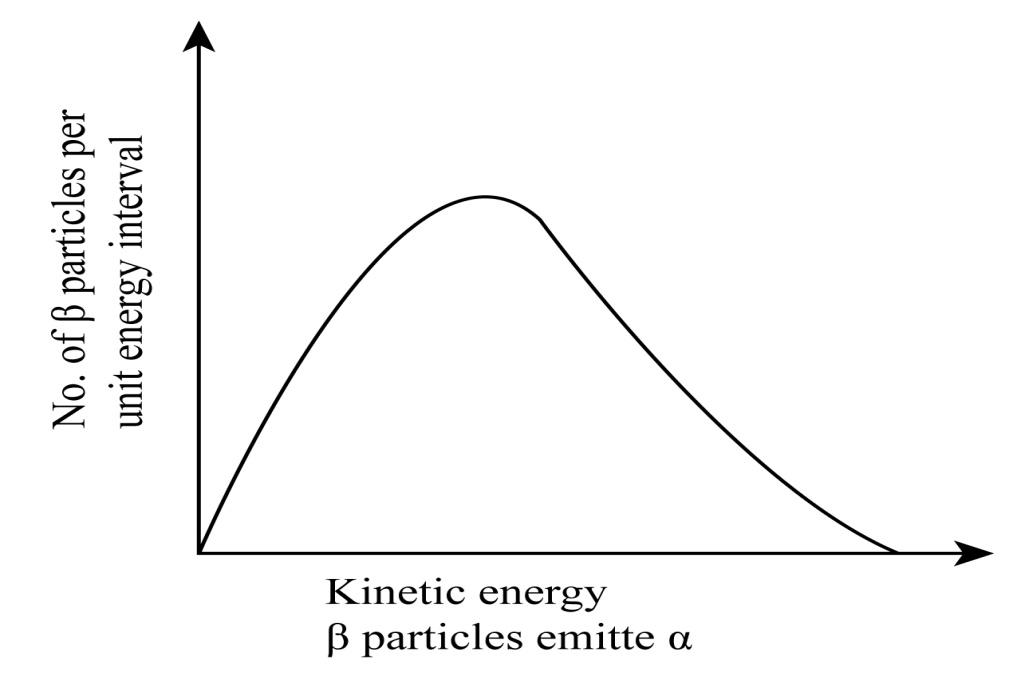
Show that the two-body decay of this type must necessarily give an electron of fixed energy and, therefore, cannot for the observed continuous energy distribution in the $\beta $-decay of a neutron or a nucleus.

Answer
604.2k+ views
Hint: According to conservation of linear momentum, the initial linear momentum of the system is always equal to final linear momentum. Also, the energy of the system should also remain conserved as per the law of conservation of energy.
Complete step by step answer:
Given: Neutron particle decays into two particles: a proton and an electron.
The formula to calculate the initial momentum of neutron is
$ {p_i} = {m_n}{v_i}$
Here, ${p_i}$ is the initial momentum, ${m_n}$ is the mass of the neutron and ${v_i}$ is the initial velocity.
Initially the neutron is at rest therefore the initial velocity of the neutron is zero.
Substitute$0$for ${v_i}$ in the formula to calculate the initial momentum of the neutron.
$\begin{array}{c}
{p_i} = {m_n}\left( 0 \right)\\
= 0
\end{array}$
The formula to calculate the final momentum of the decay particles is
${p_f} = {m_p}{v_p} + {m_e}{v_e} $
Here, ${p_f}$ is the final momentum,${m_p}$ is the mass of proton, ${v_p}$ is the velocity of proton, ${m_e}$ is the mass of electron and ${v_e}$ is the velocity of electron.
According to conservation of linear momentum,
${p_i} = {p_f} $
Substitute $0$ for ${p_i}$ and ${m_p}{v_p} + {m_e}{v_e}$ for ${p_f}$ in the conservation of energy and solve to obtain the formula for velocity of proton.
$ 0 = {m_p}{v_p} + {m_e}{v_e}\\$
$\implies {m_p}{v_p} = - {m_e}{v_e}\\$
$\implies {v_p} = - \dfrac{{{m_e}{v_e}}}{{{m_p}}}$
This negative sign in the value of velocity shows that the proton and electron moves in the opposite direction with respect to each other.
Initially as the neutron is at rest, it means the neutron possesses some rest mass energy. The formula to calculate the rest mass energy of the neutron is
${E_n} = \Delta m{c^2}$
Here, ${E_n}$ is the rest mass energy of the neutron, $\Delta m$ is the mass converted into energy and $c$ is the speed of light.
After the decay, both the particles possess kinetic energy. The formula to calculate the total energy of the system after decay is
${E_f} = \dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}{m_p}v_p^2$
Here, ${E_f}$ is the final energy of the system.
According to conservation of energy,
${E_n} = {E_f}$
Substitute $\Delta m{c^2}$ for${E_n}$ and$\dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}{m_p}v_p^2$ for ${E_f}$ in the conservation of energy.
$\Delta m{c^2} = \dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}{m_p}v_p^2$
Substitute$ - \dfrac{{{m_e}{v_e}}}{{{m_p}}}$ for${v_p}$ in the above equation and solve to calculate the velocity of the electron.
$ \Delta m{c^2} = \dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}{m_p}{\left( { - \dfrac{{{m_e}{v_e}}}{{{m_p}}}} \right)^2}\\$
$\implies \Delta m{c^2} = \dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}\dfrac{{m_e^2v_e^2}}{{{m_p}}}\\$
$\implies \dfrac{{v_e^2}}{2}{m_e}\left( {1 + \dfrac{{{m_e}}}{{{m_p}}}} \right) = \Delta m{c^2}\\$
$\therefore v_e^2 = \dfrac{{2\Delta m{c^2}}}{{{m_e}\left( {1 + \dfrac{{{m_e}}}{{{m_p}}}} \right)}}$
This shows that the velocity of the electron is always a fixed value; therefore the energy of the electron should be a fixed value. This is the reason why a continuous energy distribution cannot be observed.
Note:
The simple result for this exercise was one among the several arguments advanced by W.Pauli to predict the existence of a third particle in the decay products of $\beta $-decay. This particle is known as neutrino. We now know that it is a particle of intrinsic spin ½ (like p, e or n), but is neutral, and either massless or having an extremely small mass (compared to the mass of an electron) and which interacts very weakly with matter. The correct decay process of the neutron is: $n \to p + {e^ - } + v$ ]
Complete step by step answer:
Given: Neutron particle decays into two particles: a proton and an electron.
The formula to calculate the initial momentum of neutron is
$ {p_i} = {m_n}{v_i}$
Here, ${p_i}$ is the initial momentum, ${m_n}$ is the mass of the neutron and ${v_i}$ is the initial velocity.
Initially the neutron is at rest therefore the initial velocity of the neutron is zero.
Substitute$0$for ${v_i}$ in the formula to calculate the initial momentum of the neutron.
$\begin{array}{c}
{p_i} = {m_n}\left( 0 \right)\\
= 0
\end{array}$
The formula to calculate the final momentum of the decay particles is
${p_f} = {m_p}{v_p} + {m_e}{v_e} $
Here, ${p_f}$ is the final momentum,${m_p}$ is the mass of proton, ${v_p}$ is the velocity of proton, ${m_e}$ is the mass of electron and ${v_e}$ is the velocity of electron.
According to conservation of linear momentum,
${p_i} = {p_f} $
Substitute $0$ for ${p_i}$ and ${m_p}{v_p} + {m_e}{v_e}$ for ${p_f}$ in the conservation of energy and solve to obtain the formula for velocity of proton.
$ 0 = {m_p}{v_p} + {m_e}{v_e}\\$
$\implies {m_p}{v_p} = - {m_e}{v_e}\\$
$\implies {v_p} = - \dfrac{{{m_e}{v_e}}}{{{m_p}}}$
This negative sign in the value of velocity shows that the proton and electron moves in the opposite direction with respect to each other.
Initially as the neutron is at rest, it means the neutron possesses some rest mass energy. The formula to calculate the rest mass energy of the neutron is
${E_n} = \Delta m{c^2}$
Here, ${E_n}$ is the rest mass energy of the neutron, $\Delta m$ is the mass converted into energy and $c$ is the speed of light.
After the decay, both the particles possess kinetic energy. The formula to calculate the total energy of the system after decay is
${E_f} = \dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}{m_p}v_p^2$
Here, ${E_f}$ is the final energy of the system.
According to conservation of energy,
${E_n} = {E_f}$
Substitute $\Delta m{c^2}$ for${E_n}$ and$\dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}{m_p}v_p^2$ for ${E_f}$ in the conservation of energy.
$\Delta m{c^2} = \dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}{m_p}v_p^2$
Substitute$ - \dfrac{{{m_e}{v_e}}}{{{m_p}}}$ for${v_p}$ in the above equation and solve to calculate the velocity of the electron.
$ \Delta m{c^2} = \dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}{m_p}{\left( { - \dfrac{{{m_e}{v_e}}}{{{m_p}}}} \right)^2}\\$
$\implies \Delta m{c^2} = \dfrac{1}{2}{m_e}v_e^2 + \dfrac{1}{2}\dfrac{{m_e^2v_e^2}}{{{m_p}}}\\$
$\implies \dfrac{{v_e^2}}{2}{m_e}\left( {1 + \dfrac{{{m_e}}}{{{m_p}}}} \right) = \Delta m{c^2}\\$
$\therefore v_e^2 = \dfrac{{2\Delta m{c^2}}}{{{m_e}\left( {1 + \dfrac{{{m_e}}}{{{m_p}}}} \right)}}$
This shows that the velocity of the electron is always a fixed value; therefore the energy of the electron should be a fixed value. This is the reason why a continuous energy distribution cannot be observed.
Note:
The simple result for this exercise was one among the several arguments advanced by W.Pauli to predict the existence of a third particle in the decay products of $\beta $-decay. This particle is known as neutrino. We now know that it is a particle of intrinsic spin ½ (like p, e or n), but is neutral, and either massless or having an extremely small mass (compared to the mass of an electron) and which interacts very weakly with matter. The correct decay process of the neutron is: $n \to p + {e^ - } + v$ ]
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




