Complete the following reaction equation :
A. \[{C_6}{H_5}{N_2}Cl + {H_3}P{O_2} + {H_2}O \to \]
B. ${C_6}{H_5}N{H_2} + B{r_2}\left( {aq.} \right) \to $
Answer
607.8k+ views
Hint: ${C_6}{H_5}{N_2}Cl$ is known as benzenediazonium-chloride. It will undergo a substitution reaction while reacting with hypophosphorous acid .
Complete step by step answer: ${C_6}{H_5}{N_2}Cl$ is known as benzenediazonium chloride. It is a saet of a diazonium cation and chloride. It exists as a colourless solid.
As we know the diazonium group is a very good leaving group and being positively charged, it takes away the two electrons of the bond that it has with the phenyl group. So, the incoming group that replaces $N_2^ + $ has to be a nucleophile.
-Hence, from ${H_3}P{O_2}$ the nucleophilic group that can be formed will be ${H_3}PO_2^ - $. The presence of ${H_2}O$ in the acid can also act as a nucleophile through its oxygen.
The products formed by the reaction of benzenediazonium chloride are ${C_6}{H_6},{N_2},HCl$ and ${H_3}P{O_3}$.
The reaction equation can be written as :-
${C_6}{H_5}{N_2}Cl + {H_3}P{O_2} + {H_2}O \to {C_6}{H_6} + {N_2} + {H_3}P{O_3} + HCl$
-when ${C_6}{H_5}{N_2}Cl$ that is benzenediazonium chloride reacts with $B{r_2}$ then the product will be $2,4,6 - tribromoaniline$ the reaction equation involved is :-
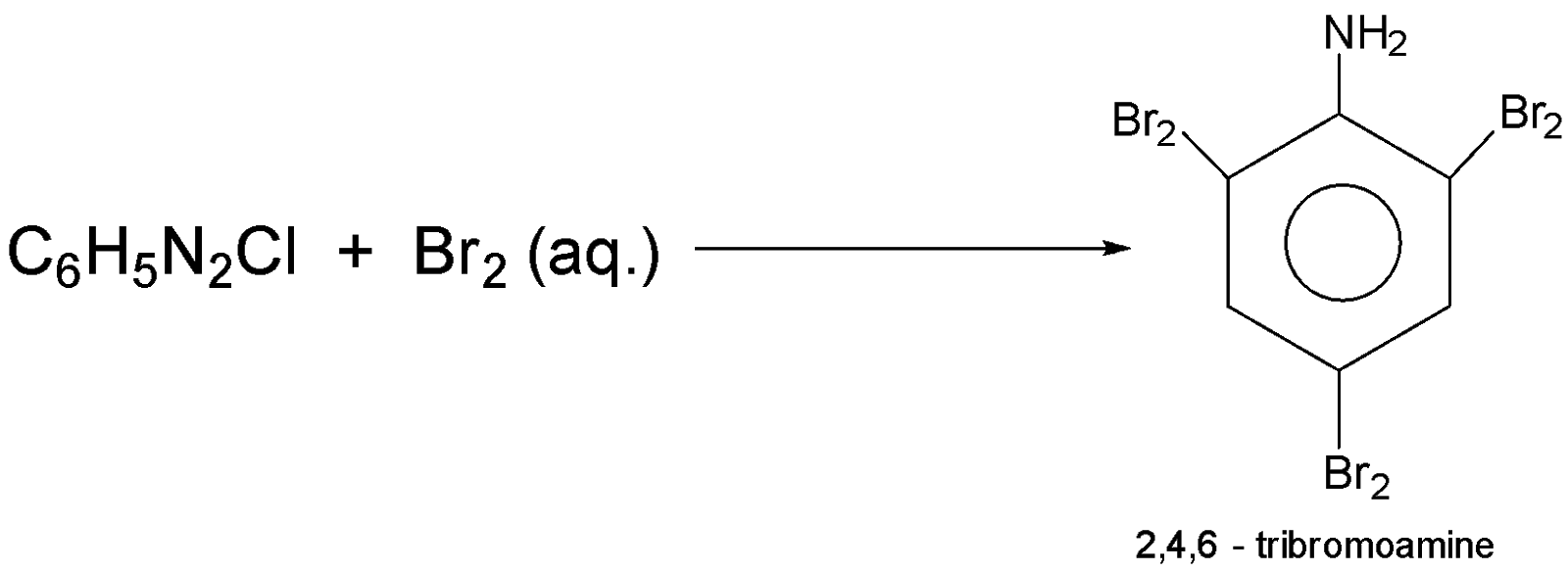
Additional Information: Benzenediazonium chloride is the parent member of the aryl diazonium compound that has use in organic chemistry; this salt is highly unstable.
Note: The diazo group $\left( {{N_2}} \right)$can be replaced with many other groups, usually anions, that give a variety of substituted phenyl derivatives.
Moreover, this compound seems to be explosive.
Complete step by step answer: ${C_6}{H_5}{N_2}Cl$ is known as benzenediazonium chloride. It is a saet of a diazonium cation and chloride. It exists as a colourless solid.
As we know the diazonium group is a very good leaving group and being positively charged, it takes away the two electrons of the bond that it has with the phenyl group. So, the incoming group that replaces $N_2^ + $ has to be a nucleophile.
-Hence, from ${H_3}P{O_2}$ the nucleophilic group that can be formed will be ${H_3}PO_2^ - $. The presence of ${H_2}O$ in the acid can also act as a nucleophile through its oxygen.
The products formed by the reaction of benzenediazonium chloride are ${C_6}{H_6},{N_2},HCl$ and ${H_3}P{O_3}$.
The reaction equation can be written as :-
${C_6}{H_5}{N_2}Cl + {H_3}P{O_2} + {H_2}O \to {C_6}{H_6} + {N_2} + {H_3}P{O_3} + HCl$
-when ${C_6}{H_5}{N_2}Cl$ that is benzenediazonium chloride reacts with $B{r_2}$ then the product will be $2,4,6 - tribromoaniline$ the reaction equation involved is :-
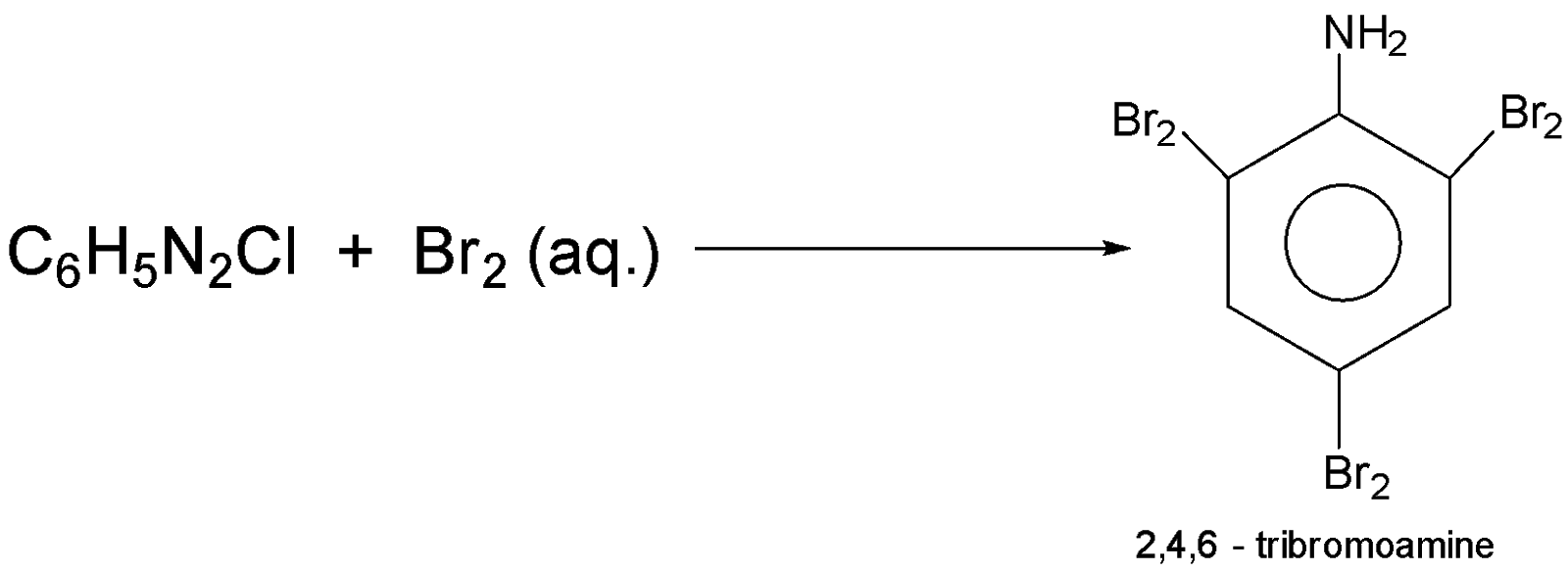
Additional Information: Benzenediazonium chloride is the parent member of the aryl diazonium compound that has use in organic chemistry; this salt is highly unstable.
Note: The diazo group $\left( {{N_2}} \right)$can be replaced with many other groups, usually anions, that give a variety of substituted phenyl derivatives.
Moreover, this compound seems to be explosive.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




