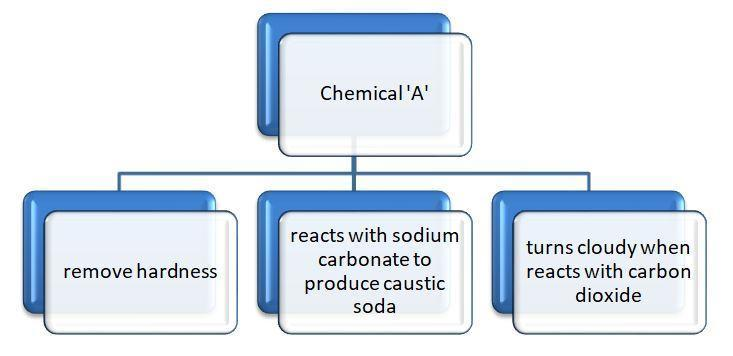
Chemical A is used for softening of water to remove temporary hardness. A reacts with sodium carbonate to produce caustic soda, when $\text{C}{{\text{O}}_{2}}$ is bubbled through ‘A’, it turns cloudy. Chemically ‘A’ is:
A. $\text{CaO}$
B. $\text{CaC}{{\text{O}}_{3}}$
C. $\text{Ca}{{\left( \text{HC}{{\text{O}}_{3}} \right)}_{2}}$
D. $\text{Ca}{{\left( \text{OH} \right)}_{2}}$
Answer
609.6k+ views
Hint: The question can be easily solved when we will draw a step by step chart or flowchart of this question.
Hardness of water is the measure of calcium and magnesium bicarbonates whose hardness can be removed by converting these bicarbonate salts into carbonates.
On reaction with sodium carbonate, it gives its corresponding carbonate.
The milkiness in the solution is due to white coloured precipitate of metal carbonate.
Complete answer: Let us see the properties of chemical ‘A’ to find the common features shown by the chemical.
Property (1)- Use to remove temporary hardness: The hardness in the water is due to high concentrations of magnesium and calcium carbonates, chlorides, bicarbonates and sulphates.
Temporary hardness (bicarbonates of calcium and magnesium) can be removed by boiling and by Clarke’s method:
Clarke's reagent is calcium hydroxide. It removes water hardness by converting bicarbonates to carbonates. The reaction is $\text{Ca}{{\left( \text{OH} \right)}_{2}}+\text{Ca}{{\left( \text{HC}{{\text{O}}_{3}} \right)}_{2}}\to 2\text{CaC}{{\text{O}}_{3}}\downarrow +2{{\text{H}}_{2}}\text{O}$.
Property (2)- Turns solution milky when reacts with $\text{C}{{\text{O}}_{2}}$ : When carbon dioxide gas is passed through lime water, it turns milky because of the formation of calcium carbonate. The reaction is $\text{Ca}{{\left( \text{OH} \right)}_{2}}\left( \text{aq}\text{.} \right)+\text{C}{{\text{O}}_{2}}\left( \text{g} \right)\to \text{CaC}{{\text{O}}_{3}}\downarrow \left( \text{s} \right)+{{\text{H}}_{2}}\text{O}\left( \text{l} \right)$.
Property (3)- Reacts with sodium carbonate to form caustic soda: Caustic soda is $\text{NaOH}$. The reaction is $\text{Ca}{{\left( \text{OH} \right)}_{2}}+\text{N}{{\text{a}}_{2}}\text{C}{{\text{O}}_{3}}\to 2\text{NaOH}+\text{CaC}{{\text{O}}_{3}}$.
The answer to this question is ‘d’. Calcium hydroxide is used to remove temporary hardness of water, reacts with sodium carbonate to form sodium hydroxide and the lime water turns milky when reacts with $\text{C}{{\text{O}}_{2}}$.
Additional Information:
Uses of calcium hydroxide:
(1) Used as a flocculant in sewage treatment plants.
(2) Used as acid neutralizer because it is a weak base and used as a pH regulator in soil.
(3) Used as filler material in rubber and plastics.
Note: The options given in the question are little confusing inter-related, as all the options are the different compounds of calcium. The point is to remember the chemical used to remove temporary hardness. The answer will be much clearer from that only. The temporary hardness is removed by slaked lime ($\text{Ca}{{\left( \text{OH} \right)}_{2}}$) only.

Hardness of water is the measure of calcium and magnesium bicarbonates whose hardness can be removed by converting these bicarbonate salts into carbonates.
On reaction with sodium carbonate, it gives its corresponding carbonate.
The milkiness in the solution is due to white coloured precipitate of metal carbonate.
Complete answer: Let us see the properties of chemical ‘A’ to find the common features shown by the chemical.
Property (1)- Use to remove temporary hardness: The hardness in the water is due to high concentrations of magnesium and calcium carbonates, chlorides, bicarbonates and sulphates.
Temporary hardness (bicarbonates of calcium and magnesium) can be removed by boiling and by Clarke’s method:
Clarke's reagent is calcium hydroxide. It removes water hardness by converting bicarbonates to carbonates. The reaction is $\text{Ca}{{\left( \text{OH} \right)}_{2}}+\text{Ca}{{\left( \text{HC}{{\text{O}}_{3}} \right)}_{2}}\to 2\text{CaC}{{\text{O}}_{3}}\downarrow +2{{\text{H}}_{2}}\text{O}$.
Property (2)- Turns solution milky when reacts with $\text{C}{{\text{O}}_{2}}$ : When carbon dioxide gas is passed through lime water, it turns milky because of the formation of calcium carbonate. The reaction is $\text{Ca}{{\left( \text{OH} \right)}_{2}}\left( \text{aq}\text{.} \right)+\text{C}{{\text{O}}_{2}}\left( \text{g} \right)\to \text{CaC}{{\text{O}}_{3}}\downarrow \left( \text{s} \right)+{{\text{H}}_{2}}\text{O}\left( \text{l} \right)$.
Property (3)- Reacts with sodium carbonate to form caustic soda: Caustic soda is $\text{NaOH}$. The reaction is $\text{Ca}{{\left( \text{OH} \right)}_{2}}+\text{N}{{\text{a}}_{2}}\text{C}{{\text{O}}_{3}}\to 2\text{NaOH}+\text{CaC}{{\text{O}}_{3}}$.
The answer to this question is ‘d’. Calcium hydroxide is used to remove temporary hardness of water, reacts with sodium carbonate to form sodium hydroxide and the lime water turns milky when reacts with $\text{C}{{\text{O}}_{2}}$.
Additional Information:
Uses of calcium hydroxide:
(1) Used as a flocculant in sewage treatment plants.
(2) Used as acid neutralizer because it is a weak base and used as a pH regulator in soil.
(3) Used as filler material in rubber and plastics.
Note: The options given in the question are little confusing inter-related, as all the options are the different compounds of calcium. The point is to remember the chemical used to remove temporary hardness. The answer will be much clearer from that only. The temporary hardness is removed by slaked lime ($\text{Ca}{{\left( \text{OH} \right)}_{2}}$) only.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE

No of 5 membered ring in left Caleft EDTA right right2 class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

How many 176Omega resistors in parallel are required class 12 physics CBSE

What is corrosion Explain its advantages and disad class 12 chemistry CBSE




