: A metal crystallises in face centred cubic structure. If the edge of its unit cell is ‘a’, the closest approach between two atoms in metallic crystal will be:
(A) ${\rm{2}}\sqrt {{\rm{2a}}} $
(B) $\sqrt {{\rm{2a}}} $
(C) $\dfrac{{\rm{a}}}{{\sqrt {\rm{2}} }}$
(D) ${\rm{2a}}$
Answer
547.6k+ views
Hint: We know that, in the face centred cubic structure the number of atoms present in it is four. Generally, the one atom is present or exists in the center and three atoms present at the corner of the FCC structure. The FCC is one of the abbreviations used for representing the face centred cubic.
Complete step-by-step solution:As we all know, for FCC crystal or face cubic structure, the closet method between two atoms in metallic crystal will be equal to the half the length of the face diagonal. It generally be represented as $\dfrac{1}{2} \times {\rm{length}}\;{\rm{of}}\;{\rm{diagonal}}......\left( 1 \right)$.
But the length of the face diagonal of the face cubic structure is $\sqrt {{\rm{2a}}} ......\left( 2 \right)$.
Where, ‘a’ is the edge of the unit cell.
So, equating the equations (1) and (2), for getting the closet value between the atoms in a metallic crystal as shown below.
$\dfrac{1}{2} \times \sqrt {{\rm{2a}}} = \dfrac{{\rm{a}}}{{\sqrt {\rm{2}} }}$
Thus, for “a metal crystallises in face centred cubic structure. If the edge of its unit cell is ‘a’, the closet approach between two atoms in metallic crystal” will be $\dfrac{{\rm{a}}}{{\sqrt {\rm{2}} }}$ as shown in the image below:
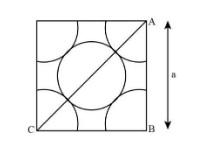
Hence, the correct option for this question is C that is $\dfrac{{\rm{a}}}{{\sqrt {\rm{2}} }}$.
Note:Usually, the crystals are divided into seven main types. Generally, it is divided on the basis of the number of atoms present in the crystals at the corner and at the center and the arrangement of atoms or ions in the crystal. The process is normally termed as crystallography.
Complete step-by-step solution:As we all know, for FCC crystal or face cubic structure, the closet method between two atoms in metallic crystal will be equal to the half the length of the face diagonal. It generally be represented as $\dfrac{1}{2} \times {\rm{length}}\;{\rm{of}}\;{\rm{diagonal}}......\left( 1 \right)$.
But the length of the face diagonal of the face cubic structure is $\sqrt {{\rm{2a}}} ......\left( 2 \right)$.
Where, ‘a’ is the edge of the unit cell.
So, equating the equations (1) and (2), for getting the closet value between the atoms in a metallic crystal as shown below.
$\dfrac{1}{2} \times \sqrt {{\rm{2a}}} = \dfrac{{\rm{a}}}{{\sqrt {\rm{2}} }}$
Thus, for “a metal crystallises in face centred cubic structure. If the edge of its unit cell is ‘a’, the closet approach between two atoms in metallic crystal” will be $\dfrac{{\rm{a}}}{{\sqrt {\rm{2}} }}$ as shown in the image below:

Hence, the correct option for this question is C that is $\dfrac{{\rm{a}}}{{\sqrt {\rm{2}} }}$.
Note:Usually, the crystals are divided into seven main types. Generally, it is divided on the basis of the number of atoms present in the crystals at the corner and at the center and the arrangement of atoms or ions in the crystal. The process is normally termed as crystallography.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




