Write the name of monomers used for getting the following polymers:
(i) Bakelite (ii) Neoprene.
Answer
267.9k+ views
Hint: Polymers are the materials made up of long, repeating chains of molecules. The polymers have unique properties, depending on the type of monomers being used for the formation of polymers.
Complete step by step solution:
(i) Bakelite: It is a copolymer made up of phenol and formaldehyde. Phenol – Formaldehyde resins are obtained by the reaction of phenol and formaldehyde in the presence of a basic catalyst. The reaction involves the formation of methylene bridges in ortho, para or both ortho and para positions of phenol. When the polymerisation of these substituted phenols take place, result into the formation of a linear polymer called Novalac which is used in paints. The reaction for this polymerisation is given below,
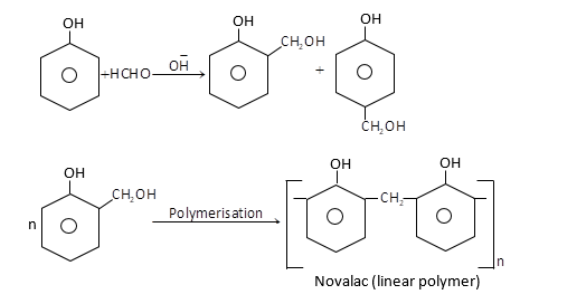
Novalac on heating with $HCHO$undergo cross linkage to form an infusible solid called bakelite.
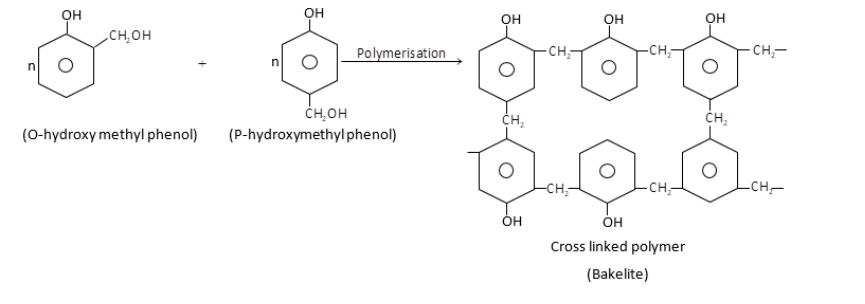
$\therefore $Monomers of bakelite are phenol and formaldehyde
(ii) Neoprene: Its monomer chloroprene is prepared from acetylene
\[\mathop {2CH \equiv CH}\limits_{acetylene} \mathop {\xrightarrow[{N{H_4}Cl}]{{C{u_2}C{l_2}}}}\limits_{\left( {343K} \right)} \mathop {{H_2}C = CH - C \equiv CH}\limits_{\left( {vinylacetylene} \right)} \xrightarrow{{HCl}}\mathop {{H_2}C = CH - C \equiv C{H_2}}\limits_{\left( {Chloroprene} \right)} \]
Chloroprene undergoes free radical polymerization to form neoprene. It polymerizes very rapidly and the reaction occurs by $1,{\text{ 4}}$- addition of one chloroprene to another.
Note: Neoprene was the first synthetic rubber manufactured on a large scale. It is also called diprene. The properties of neoprene are similar to natural rubber but neoprene is more resistant to action of oils, gasoline and other hydrocarbons. It is non – inflammable. Bakelite is used for making combs, fountain pens, electrical goods, etc. Soft Bakelite with low degree of polymerisation are used as binding glue for laminated, wooden plants and lacquers. Sulphonated bakelites are used as ion exchange resins.
Complete step by step solution:
(i) Bakelite: It is a copolymer made up of phenol and formaldehyde. Phenol – Formaldehyde resins are obtained by the reaction of phenol and formaldehyde in the presence of a basic catalyst. The reaction involves the formation of methylene bridges in ortho, para or both ortho and para positions of phenol. When the polymerisation of these substituted phenols take place, result into the formation of a linear polymer called Novalac which is used in paints. The reaction for this polymerisation is given below,

Novalac on heating with $HCHO$undergo cross linkage to form an infusible solid called bakelite.

$\therefore $Monomers of bakelite are phenol and formaldehyde
(ii) Neoprene: Its monomer chloroprene is prepared from acetylene
\[\mathop {2CH \equiv CH}\limits_{acetylene} \mathop {\xrightarrow[{N{H_4}Cl}]{{C{u_2}C{l_2}}}}\limits_{\left( {343K} \right)} \mathop {{H_2}C = CH - C \equiv CH}\limits_{\left( {vinylacetylene} \right)} \xrightarrow{{HCl}}\mathop {{H_2}C = CH - C \equiv C{H_2}}\limits_{\left( {Chloroprene} \right)} \]
Chloroprene undergoes free radical polymerization to form neoprene. It polymerizes very rapidly and the reaction occurs by $1,{\text{ 4}}$- addition of one chloroprene to another.
Note: Neoprene was the first synthetic rubber manufactured on a large scale. It is also called diprene. The properties of neoprene are similar to natural rubber but neoprene is more resistant to action of oils, gasoline and other hydrocarbons. It is non – inflammable. Bakelite is used for making combs, fountain pens, electrical goods, etc. Soft Bakelite with low degree of polymerisation are used as binding glue for laminated, wooden plants and lacquers. Sulphonated bakelites are used as ion exchange resins.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




