White phosphorus ${P_4}$ has the following characteristics.
(This question has multiple correct options)
(A) 6 P-P single bonds
(B) 4 P-P single bonds
(C) 4 lone pair of electrons
(D) P-P-P angle of ${60^\circ }$
Answer
267.9k+ views
Hint: Phosphorus is 15 group elements. Phosphorus is an essential constituent of animal and plant matter. It is present in bones as well as in living cells.
Complete step by step answer:
Phosphorus exists in several allotropic forms.
Example: White phosphorus, red phosphorus, black phosphorus, etc.
White phosphorus is in gaseous state and is a waxy solid consisting of reactive ${p_4}$tetrahedron.

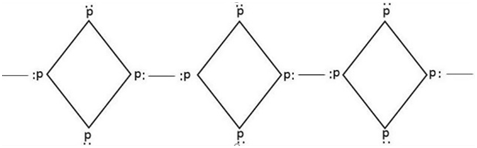
The structure of phosphorus shows 6 covalent bonds between P-atom and 4 lone pairs of electrons.
Therefore, from the above explanation, the correct option is [A] 6 p-p single bond and [C] 4 lone pair of electrons
Additional information:
White phosphorus is very reactive because ${p_4}$ rings are held together by physical bonding but no chemical bonding.
These ${p_4}$ rings are in unstable arrangement.
The red phosphorus consists of complex chain structure formed by opening of ${p_4}$cage.
Black phosphorus has a layered structure and most thermodynamically stable form.
Phosphorus can be considered as light solid.
White phosphorus is very reactive and spontaneously ignites at ${30^\circ }C$ in moist air so usually it is stored under water. So that exposure to air can be prevented.
Red phosphorus is stable at room temperature but can be converted to white phosphorus by heat, sunlight or friction.
Note: White phosphorus crystallizes in a cubic system. Phosphorus atomic size is more so it can form a single bond with other phosphorus atoms. Hence it exists as tetra atomic molecules.
Complete step by step answer:
Phosphorus exists in several allotropic forms.
Example: White phosphorus, red phosphorus, black phosphorus, etc.
White phosphorus is in gaseous state and is a waxy solid consisting of reactive ${p_4}$tetrahedron.

The structure of phosphorus shows 6 covalent bonds between P-atom and 4 lone pairs of electrons.
Therefore, from the above explanation, the correct option is [A] 6 p-p single bond and [C] 4 lone pair of electrons
Additional information:
White phosphorus is very reactive because ${p_4}$ rings are held together by physical bonding but no chemical bonding.
These ${p_4}$ rings are in unstable arrangement.
The red phosphorus consists of complex chain structure formed by opening of ${p_4}$cage.

Black phosphorus has a layered structure and most thermodynamically stable form.
Phosphorus can be considered as light solid.
White phosphorus is very reactive and spontaneously ignites at ${30^\circ }C$ in moist air so usually it is stored under water. So that exposure to air can be prevented.
Red phosphorus is stable at room temperature but can be converted to white phosphorus by heat, sunlight or friction.
Note: White phosphorus crystallizes in a cubic system. Phosphorus atomic size is more so it can form a single bond with other phosphorus atoms. Hence it exists as tetra atomic molecules.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




