Which reaction gives non resolvable products?
(A)
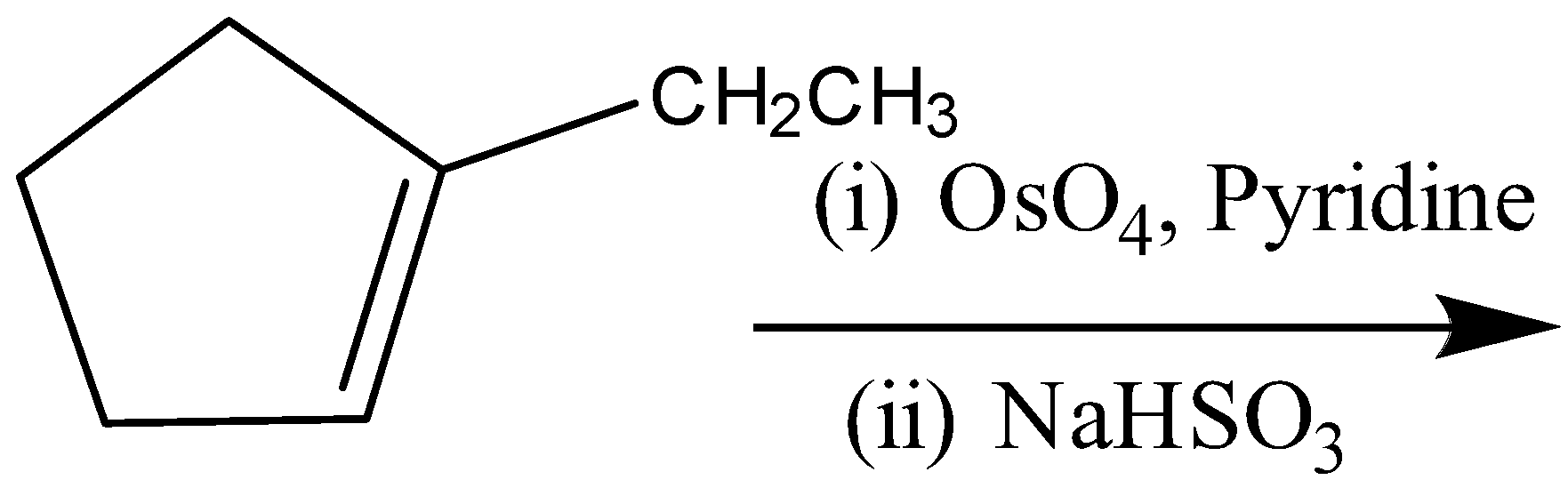
(B)
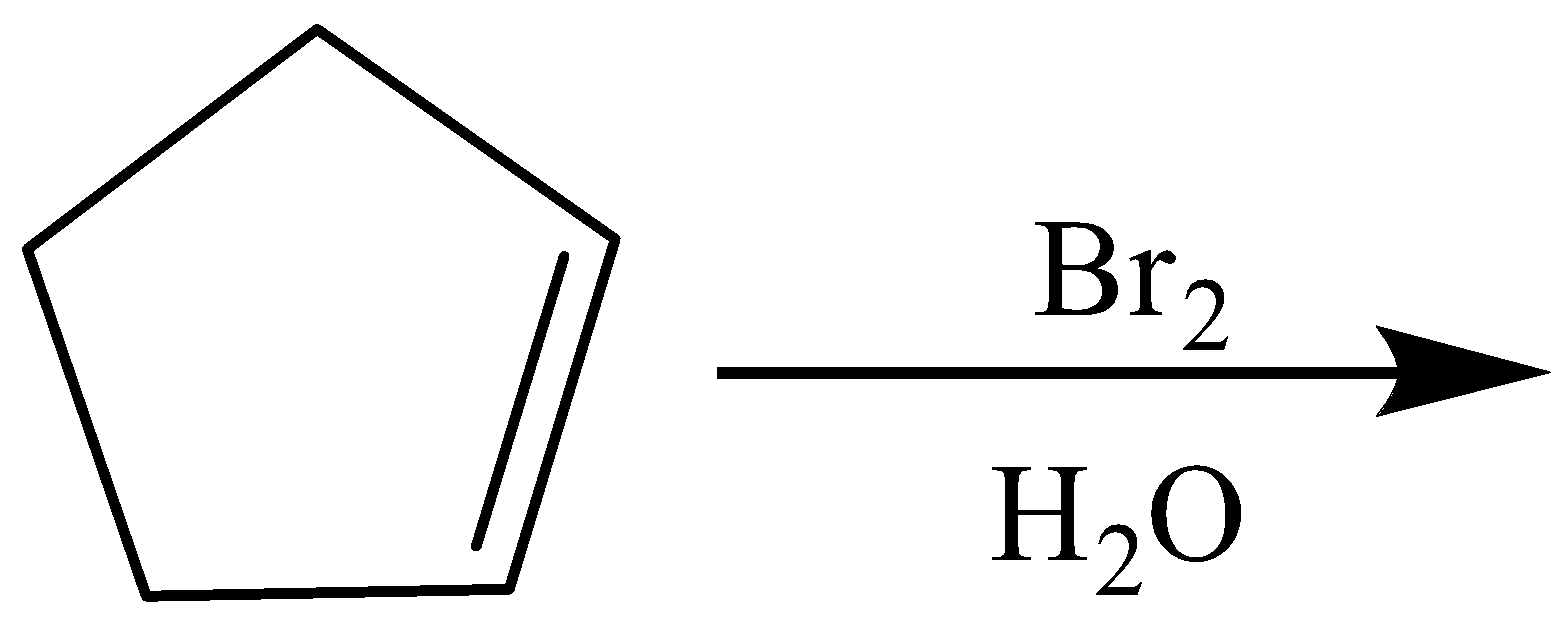
(C)
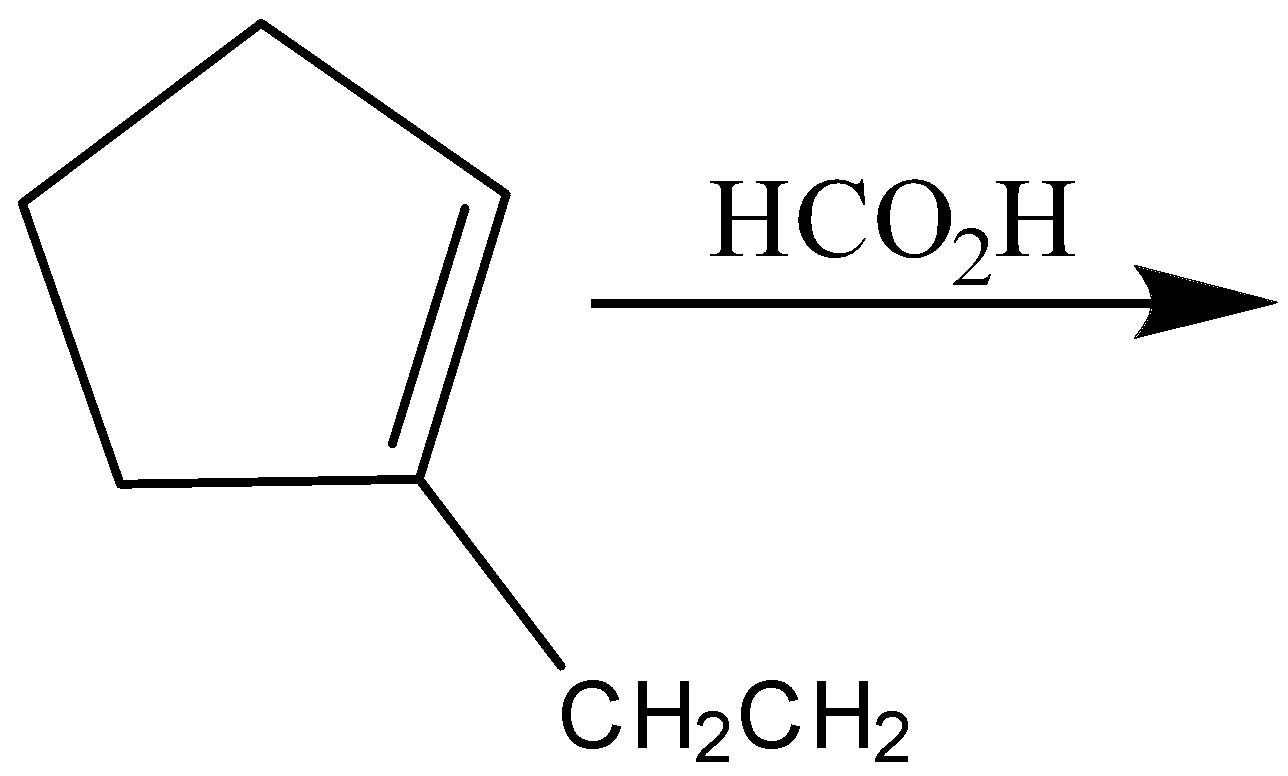
(D)
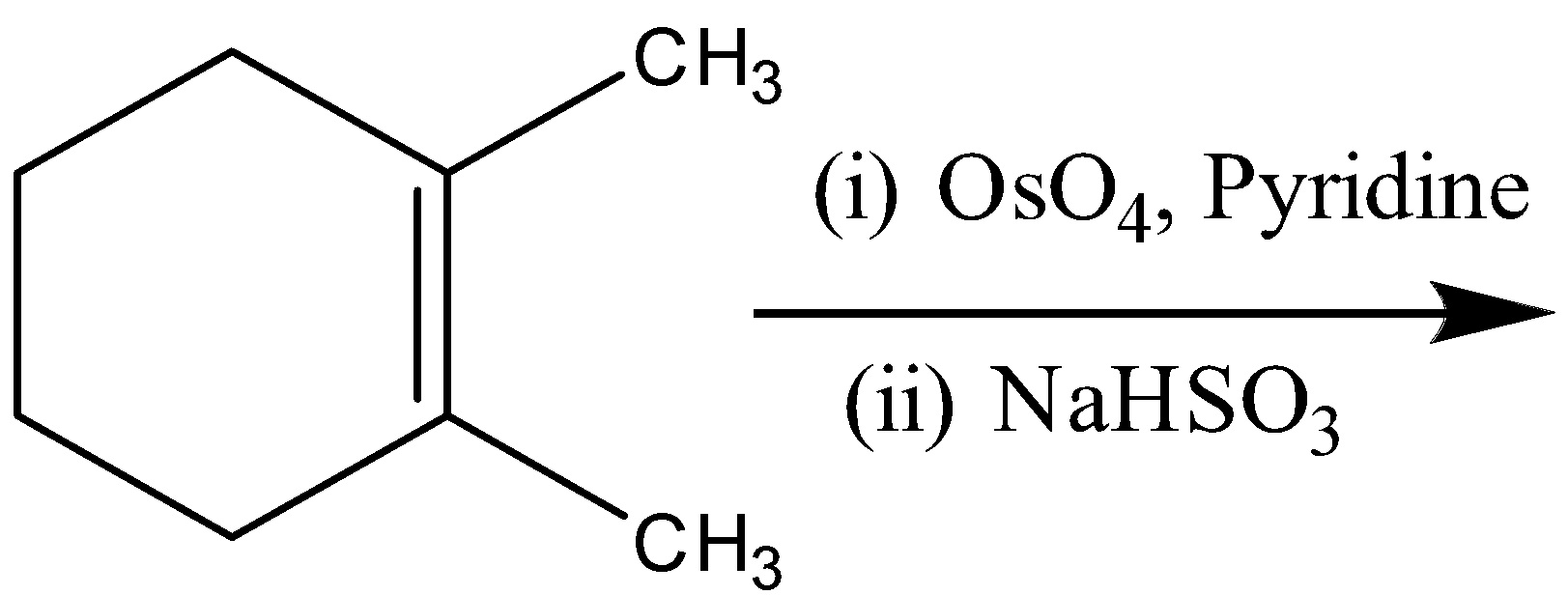
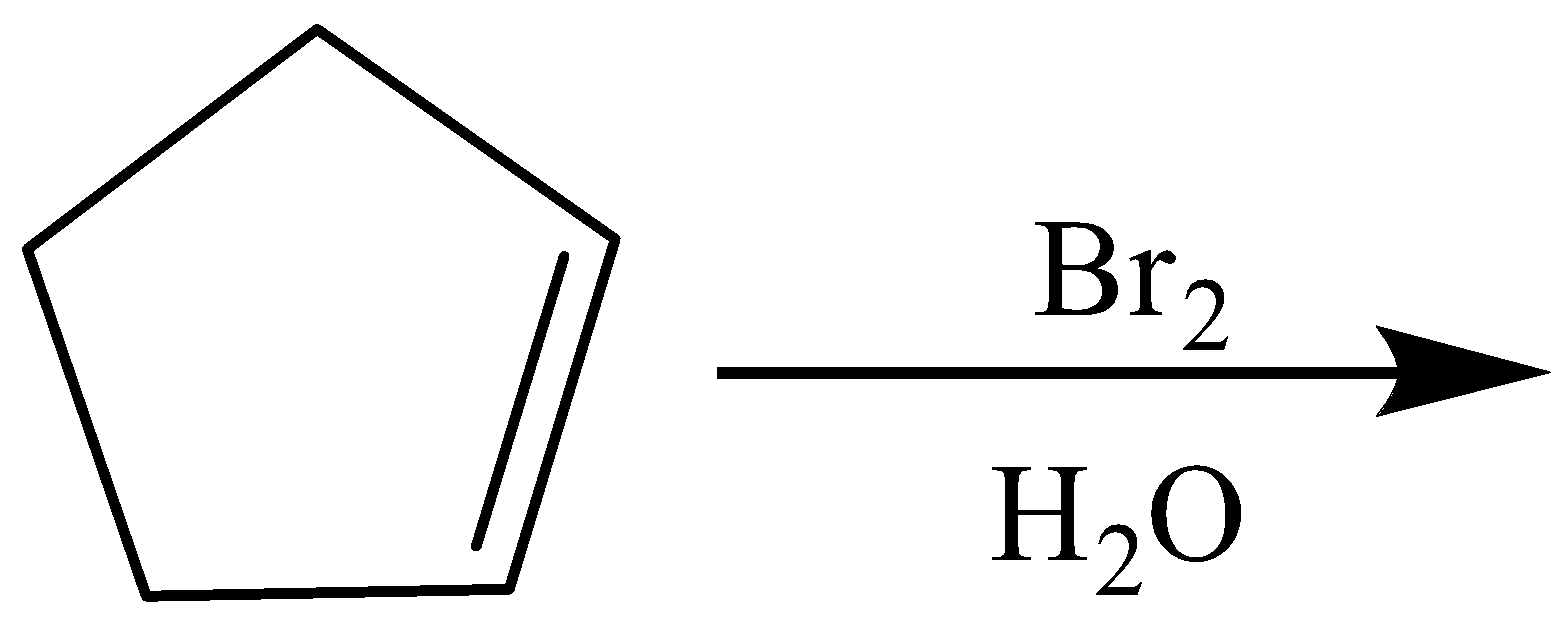

Answer
262.2k+ views
Hint: A Chiral molecule is a molecule that contains one or more carbon atoms with four different or distinct elements, and such carbon atoms are called a chiral centre or stereo genic centre or stereo centre whereas, molecules cannot he could not get through it. Their mirror image is also called a chiral molecule. The element of molecular inaccessibility in its screen is called chirality.
Complete Step by Step Solution:
The solvent compound is a compound that works visually, while the compound that is able to rotate the eyes is called optically active. All pure chiral compounds work remarkably well.
A)
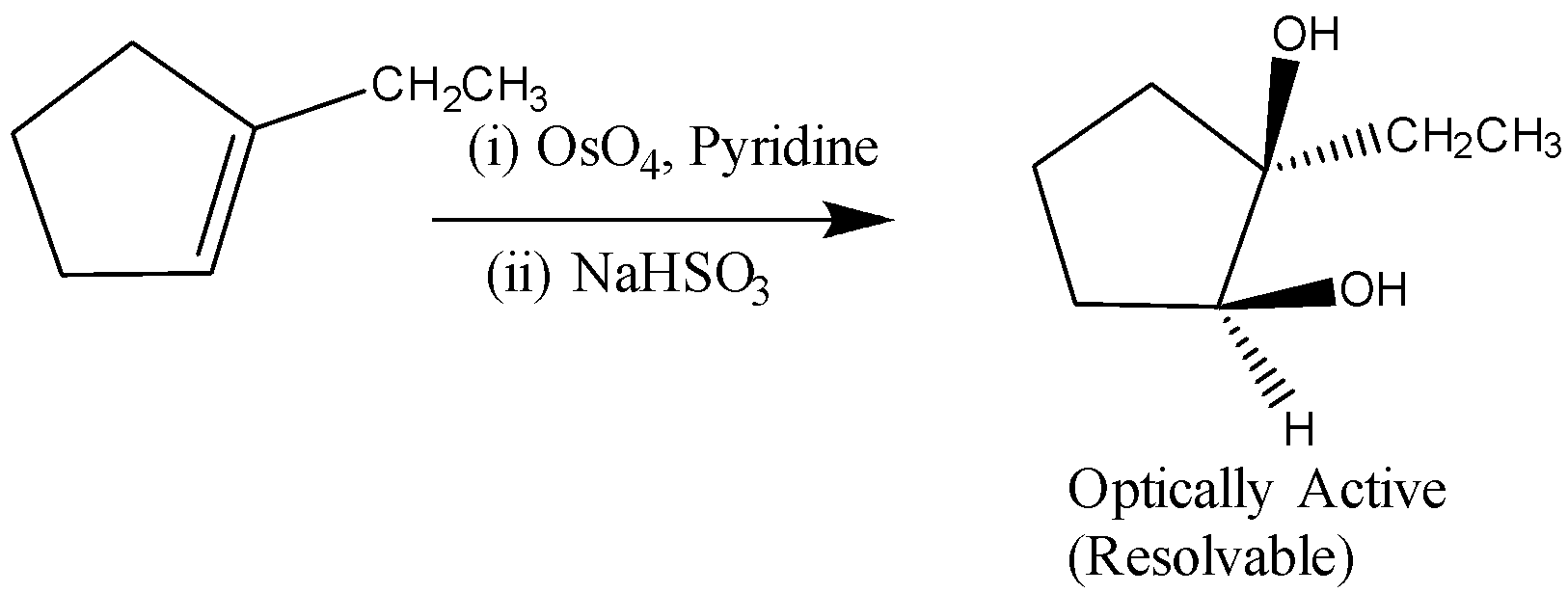
B)
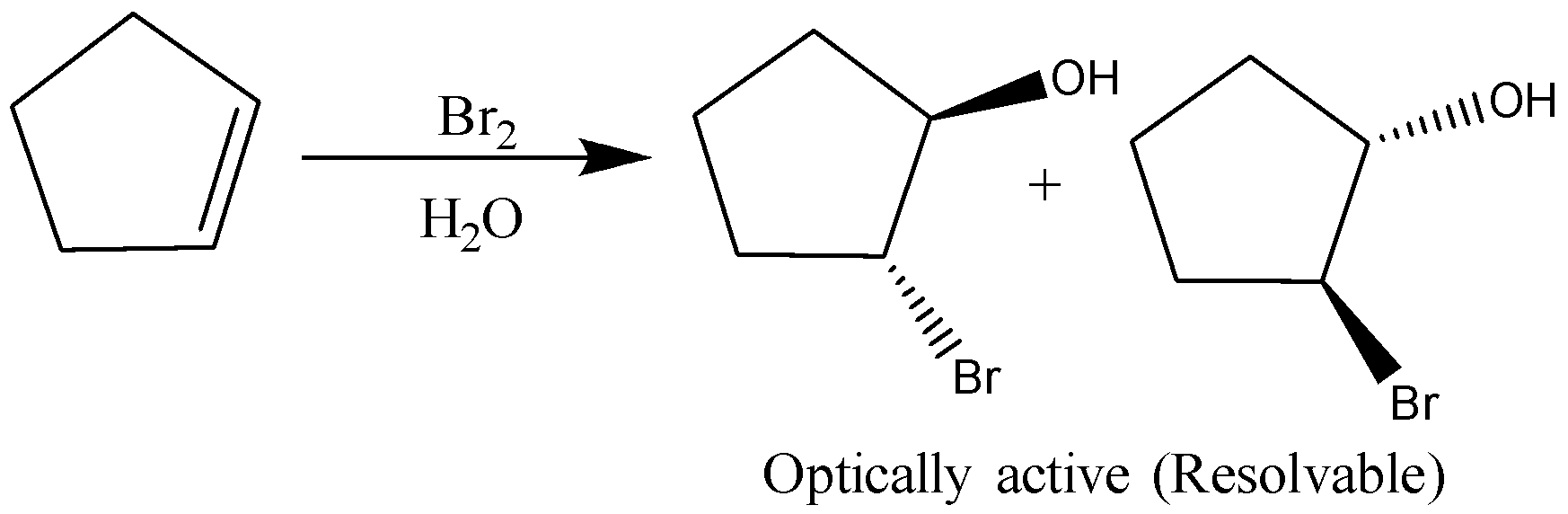
C)
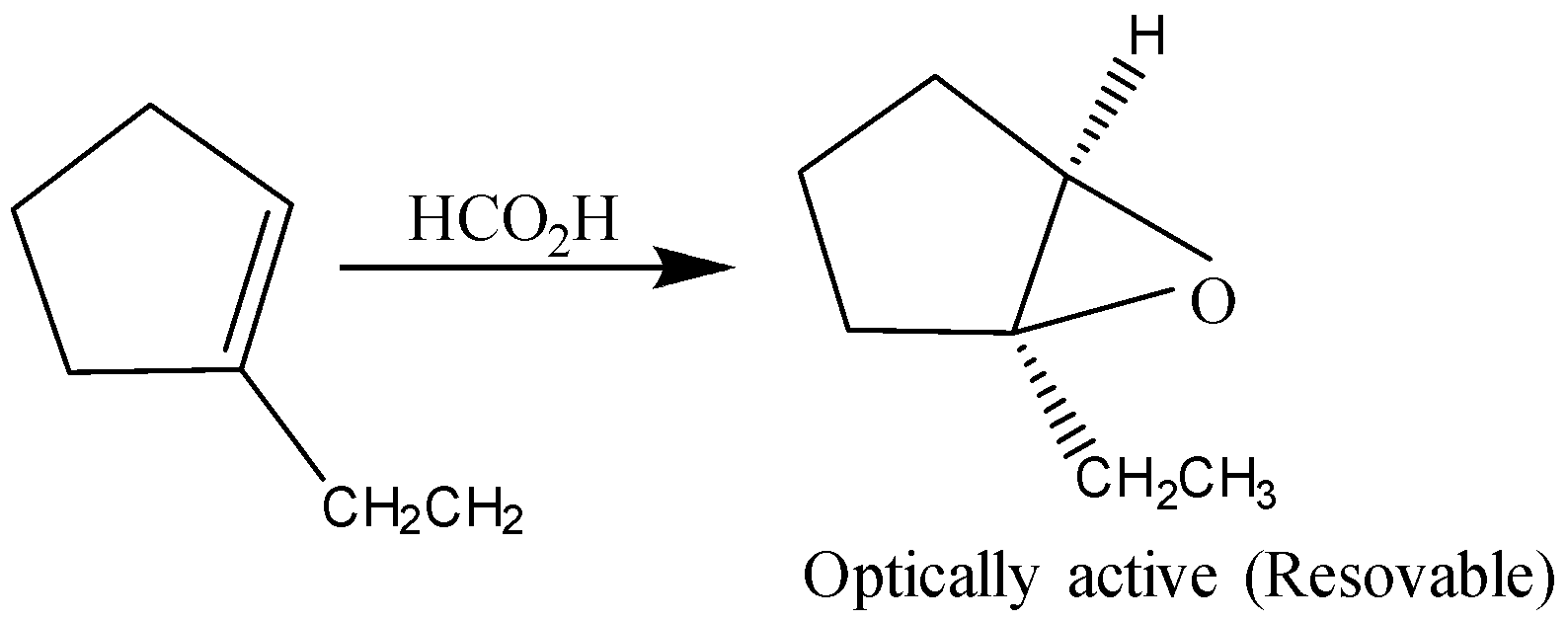
D)
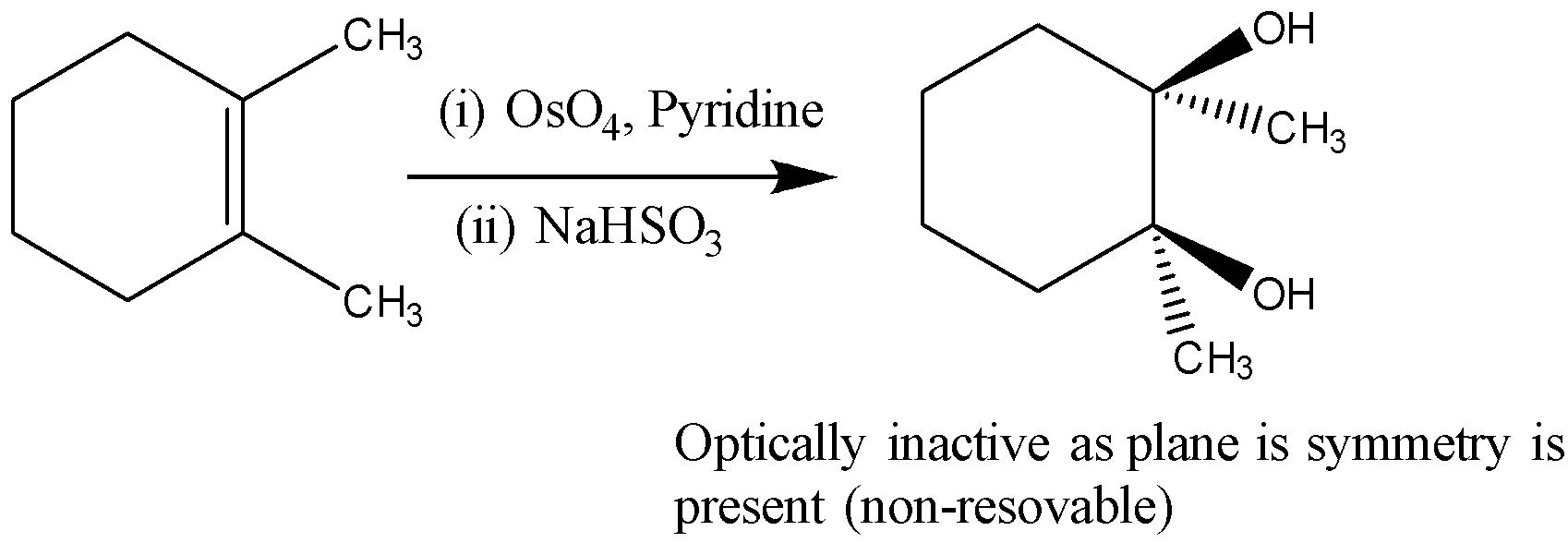
So, the correct answer is D)
Additional Information: Meso compounds will never be solved. Generally, a combination of maso must consist of two or more identical stereocenters. These molecules have an internal measuring plane that separates the compound in half. The two parts consult the inner mirror. This leads to stereochemistry being cancelled by two operators which is why it has resulted in a combination of not working visually. If A is a combination of eyes, it should have two or more stereocenters, an internal plane, and stereochemistry should be R and S.
Note: Isomer stereoisomers have the same area of atoms or groups but differ in the order of the area around the atom in the middle. Non-stereoisomers are non-mirror images of some so-called diastereomers and the combination of the eyes are those whose molecules are most powerful in their mirror images even though they contain chiral centres.
Complete Step by Step Solution:
The solvent compound is a compound that works visually, while the compound that is able to rotate the eyes is called optically active. All pure chiral compounds work remarkably well.
A)

B)

C)

D)

So, the correct answer is D)

Additional Information: Meso compounds will never be solved. Generally, a combination of maso must consist of two or more identical stereocenters. These molecules have an internal measuring plane that separates the compound in half. The two parts consult the inner mirror. This leads to stereochemistry being cancelled by two operators which is why it has resulted in a combination of not working visually. If A is a combination of eyes, it should have two or more stereocenters, an internal plane, and stereochemistry should be R and S.
Note: Isomer stereoisomers have the same area of atoms or groups but differ in the order of the area around the atom in the middle. Non-stereoisomers are non-mirror images of some so-called diastereomers and the combination of the eyes are those whose molecules are most powerful in their mirror images even though they contain chiral centres.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




