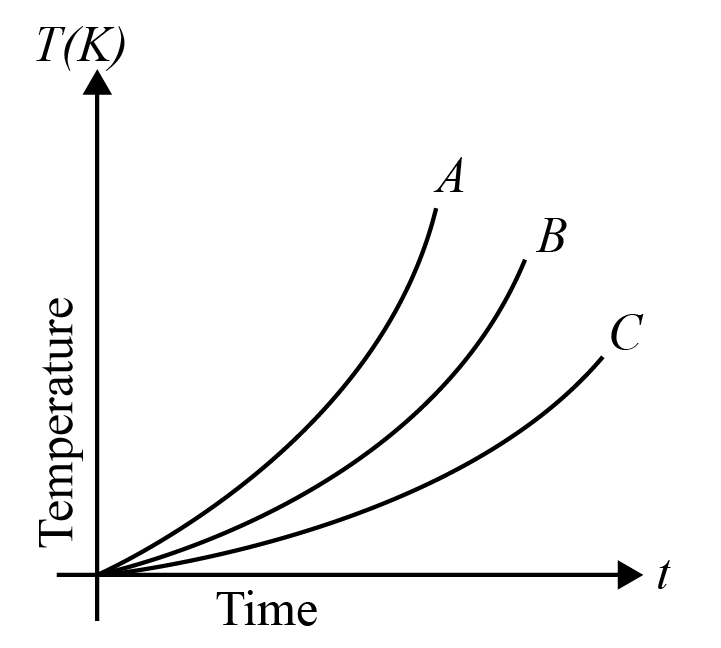
Which of the substances A, B or C has the highest specific heat? The temperature vs time graph is shown.
(A) A
(B) B
(C) C
(D) All have similar Specific heat
Answer
263.1k+ views
Hint The given graph represents 3 substances A, B , and C on their behavioral characteristics due to change in temperature. Specific heat of a substance is defined as the amount of heat required by the substance to raise the overall temperature of the substance by one degree. Apply this concept to justify your answer.
Complete Step By Step Solution
Specific heat of any substance is defined as the quantity of heat required to increase the temperature of the substance by one degree. It is also given as the ratio of heat capacity of the substance that is heated to the mass of the substance. According to this relation, we can see that specific heat is inversely proportional to the mass of the substance. But, in our given graph, the masses of the substances aren’t mentioned.
But from the same relation we can see that specific heat is directly proportional to the change in temperature of the substance. The substance which gets heated more or under application of heat for a longer period of time, is expected to have a greater specific heat quotient.
In our graph, A undergoes heating under high temperature but for rather a short period of time. Hence, its heating capacity is quite lesser than the rest of the given substances. Body B undergoes heating for a greater period of time than that of substance A, but lesser than C. Thus, body C is said to be having greater specific heat than the other substances.
Thus, Option (c) is the right answer.
Note
Specific heat of water is higher than any other substance or medium. Water plays a vital role in regulating the temperature of any substance. The specific heat quotient of water per gram is much higher than the specific heat of a metal.
Complete Step By Step Solution
Specific heat of any substance is defined as the quantity of heat required to increase the temperature of the substance by one degree. It is also given as the ratio of heat capacity of the substance that is heated to the mass of the substance. According to this relation, we can see that specific heat is inversely proportional to the mass of the substance. But, in our given graph, the masses of the substances aren’t mentioned.
But from the same relation we can see that specific heat is directly proportional to the change in temperature of the substance. The substance which gets heated more or under application of heat for a longer period of time, is expected to have a greater specific heat quotient.
In our graph, A undergoes heating under high temperature but for rather a short period of time. Hence, its heating capacity is quite lesser than the rest of the given substances. Body B undergoes heating for a greater period of time than that of substance A, but lesser than C. Thus, body C is said to be having greater specific heat than the other substances.
Thus, Option (c) is the right answer.
Note
Specific heat of water is higher than any other substance or medium. Water plays a vital role in regulating the temperature of any substance. The specific heat quotient of water per gram is much higher than the specific heat of a metal.
Recently Updated Pages
Area of an Octagon Formula Explained Simply

Absolute Pressure Formula Explained: Key Equation & Examples

Difference Between Atom and Molecule: JEE Main 2026

Difference Between Vapor and Gas: JEE Main 2026

Carbon Dioxide Formula - Definition, Uses and FAQs

Central Angle of a Circle Formula Explained Quickly

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2025-26

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics




