Which of the following is steam volatile?
(A) o-Nitrophenol
(B) p-nitrophenol
(C) Both of them
(D) Neither of them
Answer
263.1k+ views
Hint: Any compound that can be distilled with steam distillation process, is called steam volatile compound. One of the compounds from o-nitrophenol and p-nitrophenol forms a special type of bond which lowers its melting and boiling point from the other.
Complete Step-by-Step Solution:
As shown in the hint part, steam volatile compound is a compound that can be distilled by steam distillation process. Now during steam distillation, the compound needs to have its melting point under the boiling point of water so that steam will pass through the solution and the compound will melt if it is in solid state and can be separated from impurities by distillation.
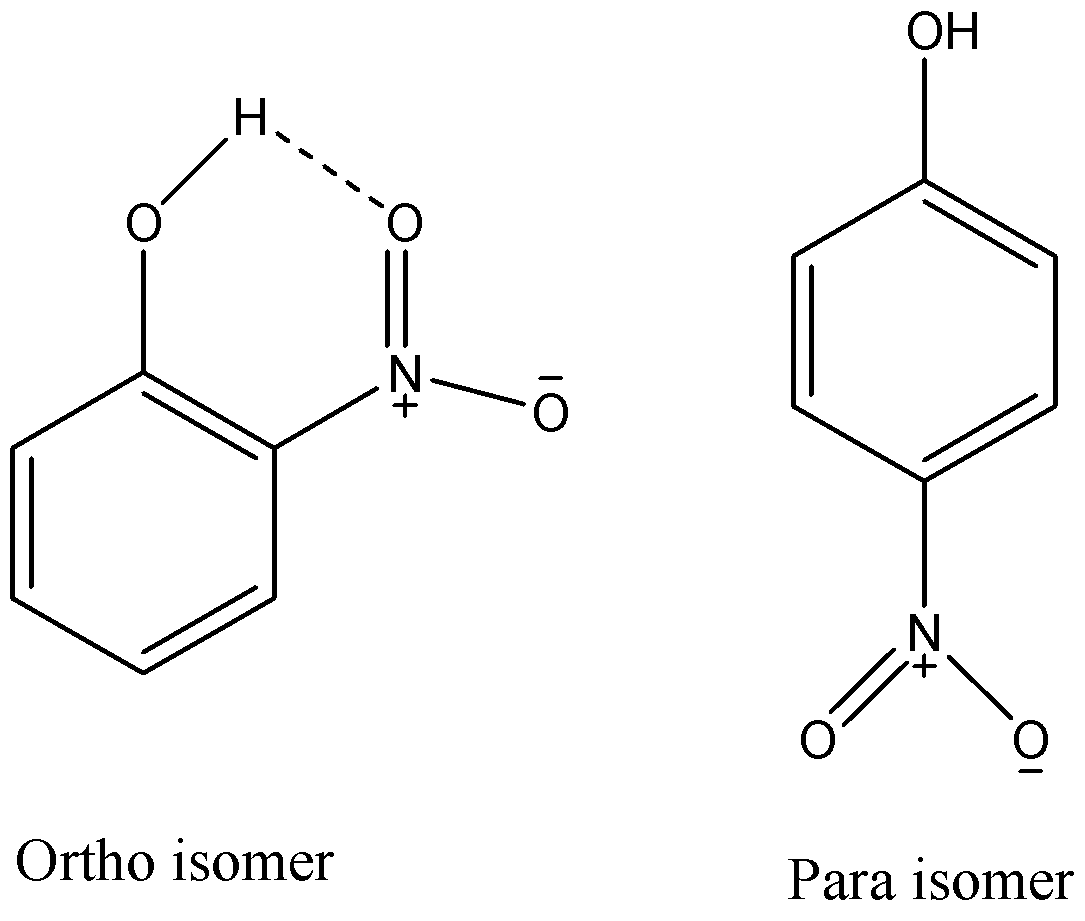
From the above given structures, we can see that ortho isomer has an intramolecular hydrogen bonding while para isomer cannot show it. As hydrogen atoms of o-nitrophenol are involved in intramolecular hydrogen bonding, they cannot effectively form hydrogen bonds with other molecules and hence it will have lower melting and boiling point then the para-isomer.
It is also practically proved that o-nitrophenol has M.P of \[{45^ \circ }C\] and p-nitrophenol has M.P of \[{119^ \circ }C\]. So, as o-nitrophenol has a lower melting point, it will easily melt and get converted to its vapours to get distilled by steam. While p-nitrophenol has a melting point higher than water’s boiling point and hence it will not be distilled with steam distillation. So, we can say that only o-nitrophenol is steam volatile.
Hence correct answer is (A) o-Nitrophenol
Note: Remember that volatile compound and steam volatile compounds are totally different terminologies. Do not get confused with intramolecular and intermolecular hydrogen bonding as both are different and have different effects on the melting point of a compound.
Complete Step-by-Step Solution:
As shown in the hint part, steam volatile compound is a compound that can be distilled by steam distillation process. Now during steam distillation, the compound needs to have its melting point under the boiling point of water so that steam will pass through the solution and the compound will melt if it is in solid state and can be separated from impurities by distillation.

From the above given structures, we can see that ortho isomer has an intramolecular hydrogen bonding while para isomer cannot show it. As hydrogen atoms of o-nitrophenol are involved in intramolecular hydrogen bonding, they cannot effectively form hydrogen bonds with other molecules and hence it will have lower melting and boiling point then the para-isomer.
It is also practically proved that o-nitrophenol has M.P of \[{45^ \circ }C\] and p-nitrophenol has M.P of \[{119^ \circ }C\]. So, as o-nitrophenol has a lower melting point, it will easily melt and get converted to its vapours to get distilled by steam. While p-nitrophenol has a melting point higher than water’s boiling point and hence it will not be distilled with steam distillation. So, we can say that only o-nitrophenol is steam volatile.
Hence correct answer is (A) o-Nitrophenol
Note: Remember that volatile compound and steam volatile compounds are totally different terminologies. Do not get confused with intramolecular and intermolecular hydrogen bonding as both are different and have different effects on the melting point of a compound.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




