Which of the following figures represents the cross-section of an octahedral site-
A)
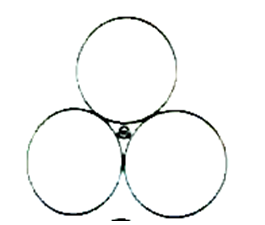
B)
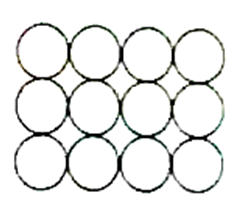
C)
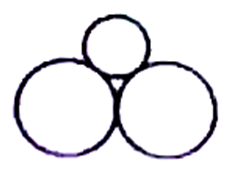
D)
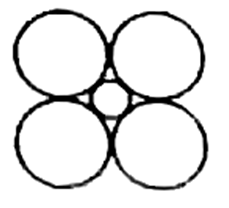
Answer
261k+ views
Hint: The vacuum created by equilateral triangles with alternate apices is known as an "octahedral void." Tetrahedral voids are the empty spaces between four spheres that are arranged in a tetrahedral pattern.
Complete Step by Step Solution:
A place at the core of a cluster of six atoms that make up an octahedron is known as an octahedral site. In comparison to the tetrahedral site, the octahedral space is bigger. There is one octahedral site for each atom when atoms of the same size are grouped as tightly as feasible.
Consider this figure
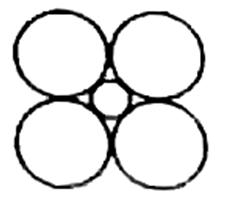
The cross section of an octahedral site is shown in this illustration. Octahedral layer refers to the interstitial void created by the union of two triangular voids from the first and second layers.
So the correct answer is option (D).
Note: We must keep in mind that the voids, which exist in crystal structures as empty spaces, are caused by the different atomic configurations. Tetrahedral and octahedral voids are the two basic categories of voids. Tetrahedral and octahedral voids differ in that tetrahedral void can only be observed in materials with tetrahedral crystal systems, whereas octahedral void can only be observed in materials with octahedral crystal systems. When six atoms, groups of atoms, or ligands are symmetrically positioned around a central atom to form the vertices of an octahedron, the shape of the compound is called octahedral molecular geometry. The prefix octa refers to the octahedron, which has eight faces. Although octahedral compounds typically have an atom in their centre and no links between the ligand atoms, the octahedron is one of the Platonic solids.
Complete Step by Step Solution:
A place at the core of a cluster of six atoms that make up an octahedron is known as an octahedral site. In comparison to the tetrahedral site, the octahedral space is bigger. There is one octahedral site for each atom when atoms of the same size are grouped as tightly as feasible.
Consider this figure

The cross section of an octahedral site is shown in this illustration. Octahedral layer refers to the interstitial void created by the union of two triangular voids from the first and second layers.
So the correct answer is option (D).
Note: We must keep in mind that the voids, which exist in crystal structures as empty spaces, are caused by the different atomic configurations. Tetrahedral and octahedral voids are the two basic categories of voids. Tetrahedral and octahedral voids differ in that tetrahedral void can only be observed in materials with tetrahedral crystal systems, whereas octahedral void can only be observed in materials with octahedral crystal systems. When six atoms, groups of atoms, or ligands are symmetrically positioned around a central atom to form the vertices of an octahedron, the shape of the compound is called octahedral molecular geometry. The prefix octa refers to the octahedron, which has eight faces. Although octahedral compounds typically have an atom in their centre and no links between the ligand atoms, the octahedron is one of the Platonic solids.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




