Which of the following arrangements of molecules is correct on the basis of their dipole moments?
(A) $B{F_3} > N{F_3} > N{H_3}$
(B) $N{F_3} > B{F_3} > N{H_3}$
(C) $N{H_3} > B{F_3} > N{F_3}$
(D) $N{H_3} > N{F_3} > B{F_3}$
Answer
261.3k+ views
Hint: How far away two electrical charges are from one another is measured in terms of a dipole moment. Dipole moments are a type of vector. The direction is from negative charge to positive charge, and the magnitude is equal to the charge times the distance between the charges. The formula of the dipole moment is $p = qd$ .
Complete step by step solution:
As we discussed above some characteristics of Dipole moment, let we consider the correct arrangement for the molecules on the basis of their dipole moment,
We know that, In $N{H_3}$ , the charge from three bonds was delocalised in the direction of lone pairs, towards nitrogen.
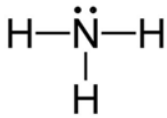
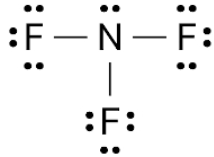
$N{F_3}$ has a greater dipole moment than $B{F_3}$ as a result of its pyramidal and trigonal planar structures. For $B{F_3}$ , there is no resulting dipole.
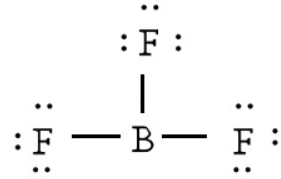
Therefore, from the above conclusion we get the correct sequence is $N{H_3} > N{F_3} > B{F_3}$ .
Hence, the correct option is (D).
Note: The dipole moment is helpful in determining how polar the chemical connection is: The bond becomes more polar as the size of the dipole moment grows. Non-polar molecules are those that have no dipole moment.
Complete step by step solution:
As we discussed above some characteristics of Dipole moment, let we consider the correct arrangement for the molecules on the basis of their dipole moment,
We know that, In $N{H_3}$ , the charge from three bonds was delocalised in the direction of lone pairs, towards nitrogen.
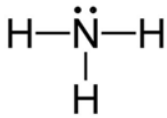

$N{F_3}$ has a greater dipole moment than $B{F_3}$ as a result of its pyramidal and trigonal planar structures. For $B{F_3}$ , there is no resulting dipole.
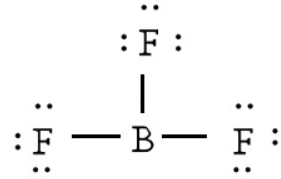
Therefore, from the above conclusion we get the correct sequence is $N{H_3} > N{F_3} > B{F_3}$ .
Hence, the correct option is (D).
Note: The dipole moment is helpful in determining how polar the chemical connection is: The bond becomes more polar as the size of the dipole moment grows. Non-polar molecules are those that have no dipole moment.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




