Urea
A.is an amide of carbonic acid
B.is diamide of carbonic acid
C.gives carbonic acid on hydrolysis
D.Resembles carbonic acid
Answer
256.2k+ views
Hint: Urea is the most widely known waste product of many living organisms. It is also a major component of human urine and animal waste. Urea is a natural product of nitrogen and protein metabolism and is prepared by heating carbon dioxide ($C{{O}_{2}}$) and ammonia ($N{{H}_{3}}$). To solve this problem first we have to know the structure of urea.
Complete answer:Urea is the major nitrogenous end product of the metabolic breakdown of proteins in human beings, animals, and some fishes. When proteins are broken down, amino groups i.e $-N{{H}_{2}}$groups are removed from an amino acid that partly comprises proteins. Further, these amino groups are converted to ammonia $(N{{H}_{3}})$. As we know ammonia is very much toxic to our body system, thereby it must be converted to urea and excreted through urine.
Urea can be made commercially by combining liquid carbon dioxide and liquid ammonia under high pressures and elevated temperatures. At first ammonium carbamate is formed which then further decomposes to urea and water at low pressures.
$\begin{align}
& 2N{{H}_{3}}+C{{O}_{2}}\to N{{H}_{2}}COON{{H}_{4}} \\
& \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(Ammonium\,\,carbamate) \\
\end{align}$
$\begin{align}
& N{{H}_{2}}COON{{H}_{4}}\to N{{H}_{2}}CON{{H}_{2}}+{{H}_{2}}O \\
& \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(Urea) \\
\end{align}$
When both the hydroxyl groups $(-OH)$ of carbonic acid (${{H}_{2}}C{{O}_{3}}$) are replaced by $-N{{H}_{2}}$ groups then the formed structure of diamide is called urea. Hence urea is a diamide of carbonic acid.
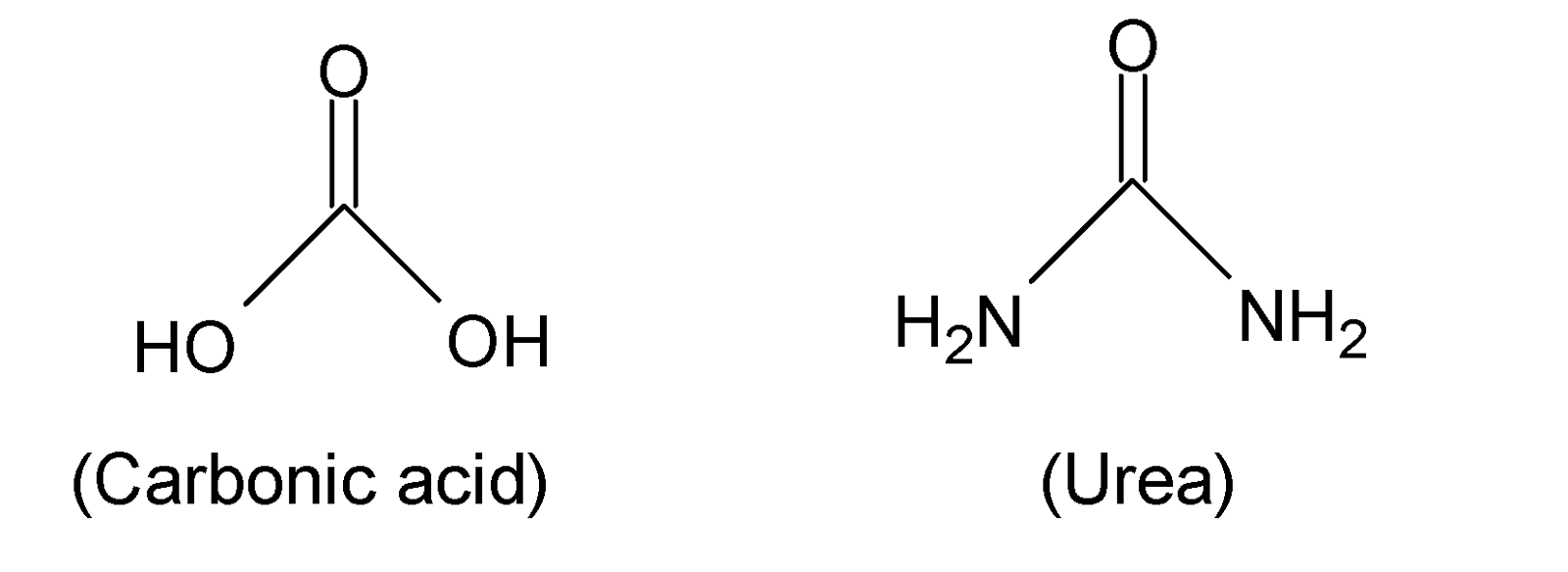
Thus, option (B) is correct.
Note: As urea contains a high amount of nitrogen, hence it can be used as fertilizer. It makes a long-lasting fertilizer and gradually releases ammonia into the soil. Nitrogen in urea is in non-protein form and hence it can be utilized in animals like sheep, and cattle. A major portion of animal protein requirements can be taken in this way.
Complete answer:Urea is the major nitrogenous end product of the metabolic breakdown of proteins in human beings, animals, and some fishes. When proteins are broken down, amino groups i.e $-N{{H}_{2}}$groups are removed from an amino acid that partly comprises proteins. Further, these amino groups are converted to ammonia $(N{{H}_{3}})$. As we know ammonia is very much toxic to our body system, thereby it must be converted to urea and excreted through urine.
Urea can be made commercially by combining liquid carbon dioxide and liquid ammonia under high pressures and elevated temperatures. At first ammonium carbamate is formed which then further decomposes to urea and water at low pressures.
$\begin{align}
& 2N{{H}_{3}}+C{{O}_{2}}\to N{{H}_{2}}COON{{H}_{4}} \\
& \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(Ammonium\,\,carbamate) \\
\end{align}$
$\begin{align}
& N{{H}_{2}}COON{{H}_{4}}\to N{{H}_{2}}CON{{H}_{2}}+{{H}_{2}}O \\
& \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(Urea) \\
\end{align}$
When both the hydroxyl groups $(-OH)$ of carbonic acid (${{H}_{2}}C{{O}_{3}}$) are replaced by $-N{{H}_{2}}$ groups then the formed structure of diamide is called urea. Hence urea is a diamide of carbonic acid.

Thus, option (B) is correct.
Note: As urea contains a high amount of nitrogen, hence it can be used as fertilizer. It makes a long-lasting fertilizer and gradually releases ammonia into the soil. Nitrogen in urea is in non-protein form and hence it can be utilized in animals like sheep, and cattle. A major portion of animal protein requirements can be taken in this way.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




