The ruff degradation used to reduce the carbon chain in an –
(A) Alcohol
(B) Alkene
(C) Ketose
(D) Aldose
Answer
268.5k+ views
Hint: Ruff degradation is a synthetic method to shorten the carbon-chain. It was first studied by Otto Ruff on glucose molecules.
Complete solution:
If we will add an aldehydic group to an acyclic form of monosaccharide, the resulting monosaccharide will be an aldose.
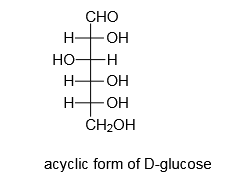
Ruff degradation will shorten the above chain by removing one carbon atom.
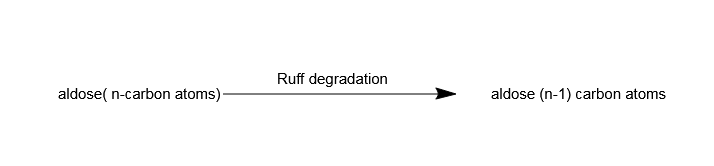
The overall conversion mechanism can be stated as follows-
The terminal aldehydic group is made to undergo selective oxidation, by using bromine water, and then it is converted to gluconate ion. In the next step, $Fe(OAc)3$ with 30% of $H2O2$ is added. This will result in the formation of $CO2$ along with a stereo selective compound from COO- ion. And in the downside, $ - CH2OH$will get converted to $ - CHO$ group. This conversion is achieved by the reduction of iron $Fe$ from its +3 state to +2 state. In this way, we get the product, D-Arabinose.
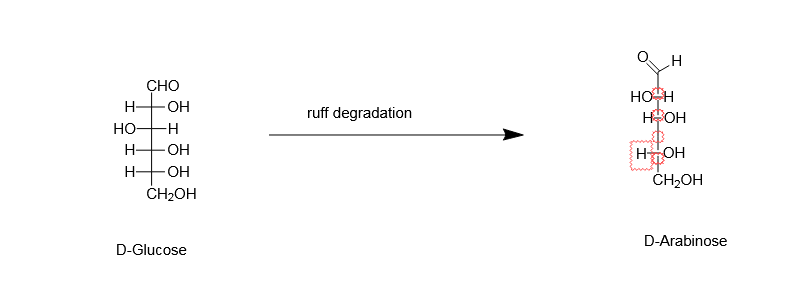
So, the correct option is D.
Note:
Ruff degradation has been named after its discoverer, Otto Ruff.
Alcohol dehydrogenase (ADH) is the enzyme that can break down alcohol into acetaldehyde. This enzyme can be found in the liver cells.
Arabinose acts as an inhibitor of enzyme Sucrase. This enzyme is present in the small intestine and helps to break down sucrose into fructose and glucose. Because of this ability, it is commercialized as a sweetener.
Arabinose is also used as a reversible switch for protein expression in E.coli
Complete solution:
If we will add an aldehydic group to an acyclic form of monosaccharide, the resulting monosaccharide will be an aldose.

Ruff degradation will shorten the above chain by removing one carbon atom.

The overall conversion mechanism can be stated as follows-
The terminal aldehydic group is made to undergo selective oxidation, by using bromine water, and then it is converted to gluconate ion. In the next step, $Fe(OAc)3$ with 30% of $H2O2$ is added. This will result in the formation of $CO2$ along with a stereo selective compound from COO- ion. And in the downside, $ - CH2OH$will get converted to $ - CHO$ group. This conversion is achieved by the reduction of iron $Fe$ from its +3 state to +2 state. In this way, we get the product, D-Arabinose.

So, the correct option is D.
Note:
Ruff degradation has been named after its discoverer, Otto Ruff.
Alcohol dehydrogenase (ADH) is the enzyme that can break down alcohol into acetaldehyde. This enzyme can be found in the liver cells.
Arabinose acts as an inhibitor of enzyme Sucrase. This enzyme is present in the small intestine and helps to break down sucrose into fructose and glucose. Because of this ability, it is commercialized as a sweetener.
Arabinose is also used as a reversible switch for protein expression in E.coli
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




