The reagent used for obtaining the osazone derivative of fructose is
A. \[N{H_2}OH\]
B. \[N{H_2}-N{H_2}\]
C. \[N{H_2}-NH{C_6}{H_5}\]
D. \[2,{\rm{ }}4 - DNP\]
Answer
267.3k+ views
Hint: Fructose, or fruit sugar, is a ketogenic simple sugar set up in numerous plants. Osazones are a category of carbohydrate derivatives formed from the reaction of reducing sugars with excess phenylhydrazine at boiling temperatures.
Complete Step by Step Answer:
Reducing sugar is any sugar that is apt to work as a reducing agent.
In an alkaline solution, a reducing sugar yields some aldehyde or ketone, which lets it work as a reducing agent, such as Benedict's reagent.
Every monosaccharide is a reducing sugar.
Fructose is a monosaccharide that has a ketone group.
So, it is a type of ketose that undergoes tautomerization to aldoses and then acts as reducing sugars.
Fructose in presence of excess phenylhydrazine forms an osazone derivative.
This reaction is a reduction of phenylhydrazine by fructose.
Fructose acts as a reducing agent by undergoing oxidation itself.
Hence, osazone is an oxidation product.
Step-1: The carbonyl group present in the fructose reacts with phenylhydrazine to form phenylhydrazone. During this phase, one molecule of hydrazine is used.
Step-2: This phenylhydrazone then reacts with two molecules of phenylhydrazine to form osazone.
The second molecule of phenylhydrazine oxidises the reactive hydroxyl group to develop the aldehyde group. This is because the ketone group had undergone a reaction in the first step itself.
Step-3: Lastly, the molecule of the phenylhydrazine that is left reacts with the newly created carbonyl group and converts fructose into osazone.
The mechanism is as follows:
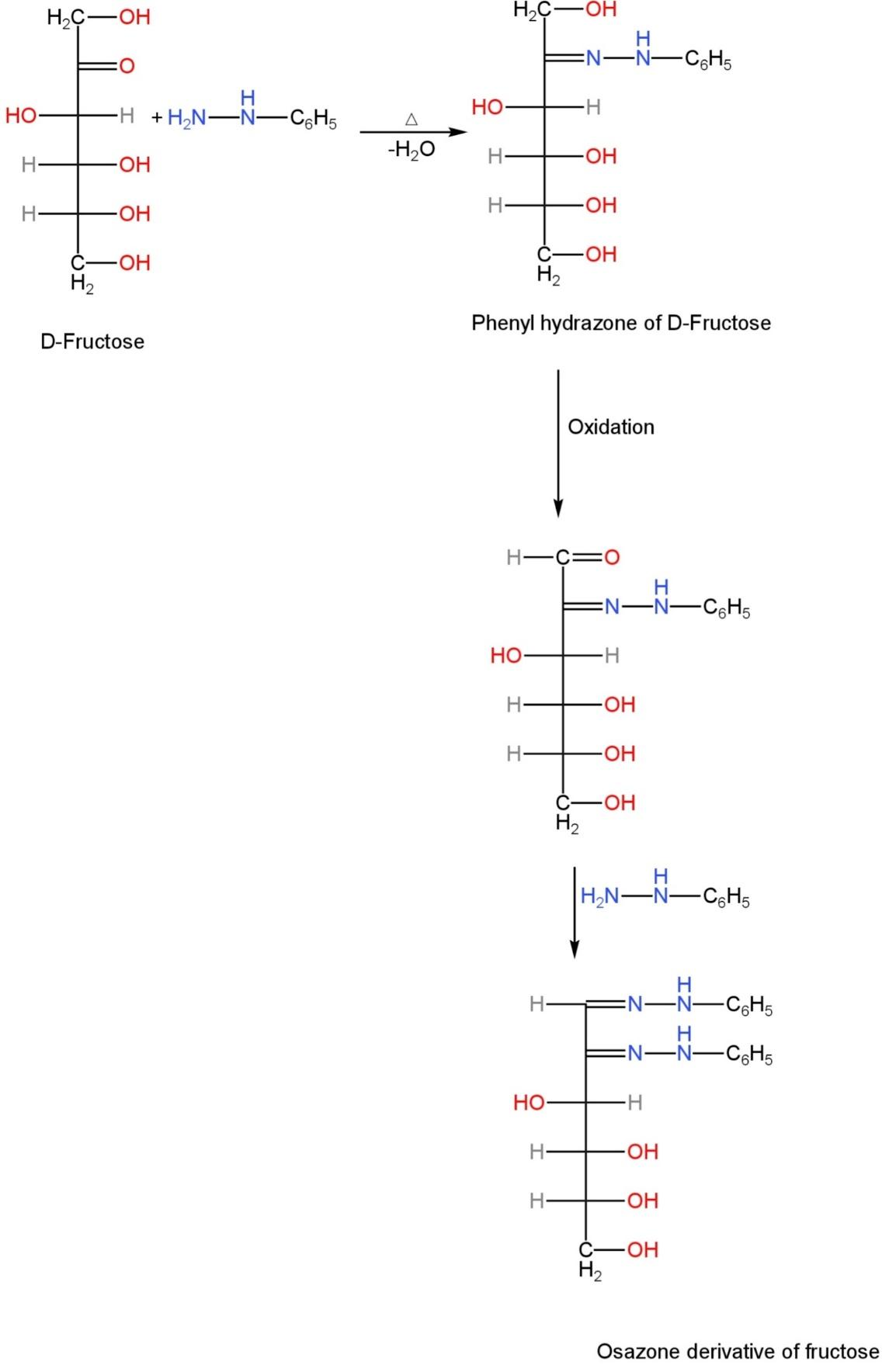
Image: Mechanism of conversion of fructose into its osazone derivative.
Out of the given options, option C i.e., \[N{H_2}-NH{C_6}{H_5}\] is the formula for phenylhydrazine.
So, option C is correct.
Note: This reaction needs a free carbonyl group. So, reducing sugars like glucose, and fructose undergoes this type of reaction. Sucrose is a non-reducing sugar that does not constitute an osazone. Sucrose is a disaccharide sugar made up of glucose and fructose subunits. It has glycosidic bonds with the carbon atoms of fructose and glucose and its transition into an open-chain form with an aldehyde group is not possible as they contain a cyclic structure.
Complete Step by Step Answer:
Reducing sugar is any sugar that is apt to work as a reducing agent.
In an alkaline solution, a reducing sugar yields some aldehyde or ketone, which lets it work as a reducing agent, such as Benedict's reagent.
Every monosaccharide is a reducing sugar.
Fructose is a monosaccharide that has a ketone group.
So, it is a type of ketose that undergoes tautomerization to aldoses and then acts as reducing sugars.
Fructose in presence of excess phenylhydrazine forms an osazone derivative.
This reaction is a reduction of phenylhydrazine by fructose.
Fructose acts as a reducing agent by undergoing oxidation itself.
Hence, osazone is an oxidation product.
Step-1: The carbonyl group present in the fructose reacts with phenylhydrazine to form phenylhydrazone. During this phase, one molecule of hydrazine is used.
Step-2: This phenylhydrazone then reacts with two molecules of phenylhydrazine to form osazone.
The second molecule of phenylhydrazine oxidises the reactive hydroxyl group to develop the aldehyde group. This is because the ketone group had undergone a reaction in the first step itself.
Step-3: Lastly, the molecule of the phenylhydrazine that is left reacts with the newly created carbonyl group and converts fructose into osazone.
The mechanism is as follows:

Image: Mechanism of conversion of fructose into its osazone derivative.
Out of the given options, option C i.e., \[N{H_2}-NH{C_6}{H_5}\] is the formula for phenylhydrazine.
So, option C is correct.
Note: This reaction needs a free carbonyl group. So, reducing sugars like glucose, and fructose undergoes this type of reaction. Sucrose is a non-reducing sugar that does not constitute an osazone. Sucrose is a disaccharide sugar made up of glucose and fructose subunits. It has glycosidic bonds with the carbon atoms of fructose and glucose and its transition into an open-chain form with an aldehyde group is not possible as they contain a cyclic structure.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




