The reaction of t-butyl bromide with sodium methoxide mainly produces:
A. isobutane
B. isobutylene
C. t-butyl methyl ether
D. sodium tert-butoxide
Answer
266.1k+ views
Hint: Think about the standard name reaction mechanism that involves the reaction of an alkyl halide and alkoxide ion. Consider mechanisms for primary, secondary, as well as tertiary alkyl halides.
Complete step by step solution:
This kind of reaction is known as the Williamson synthesis. The alkyl halide reacts with a metal alkoxide. It forms an ether is the alkyl halide is primary and an alkene if the alkyl halide is secondary or tertiary.
For a primary alkyl halide, by the $S{{N}_{2}}$ mechanism, the alkoxide ion attacks the carbon attached to the halogen atom. This halogen atom is then displaced and an ether is formed. This reaction mechanism does not favour the formation of bulky ethers. Hence, if the alkyl halide is secondary or tertiary, the reaction that happens is an elimination reaction instead of a displacement reaction due to steric hindrance.
The elimination occurs by the ${{E}_{1}}cB$ mechanism of elimination and dehydrohalogenation takes place. Thus, an alkene is formed. The reaction that occurs will be:
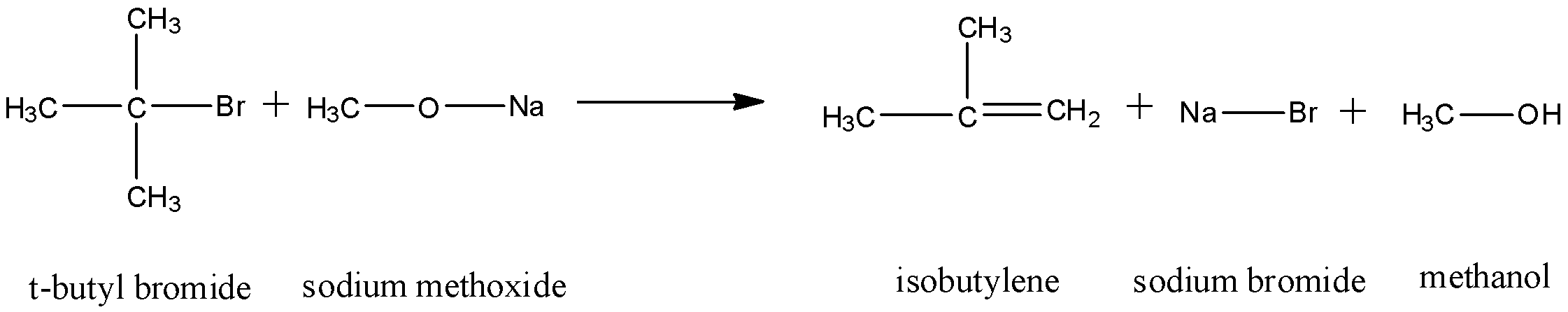
Thus, as we can see, dehydrohalogenation occurs on the tertiary carbon and an adjacent carbon. A double bond forms between them and formation of an alkene occurs.
Hence, the answer is ‘B. isobutylene’
Note: Please do not get confused due to the fact that this is a Williamson synthesis. Remember that only primary alkyl halides form ethers. Because of this, do not blindly mark your answer as ‘C. t-butyl methyl ether’. Always check if the alkyl halide is primary, secondary, or tertiary before working out the reaction and marking the answer.
Complete step by step solution:
This kind of reaction is known as the Williamson synthesis. The alkyl halide reacts with a metal alkoxide. It forms an ether is the alkyl halide is primary and an alkene if the alkyl halide is secondary or tertiary.
For a primary alkyl halide, by the $S{{N}_{2}}$ mechanism, the alkoxide ion attacks the carbon attached to the halogen atom. This halogen atom is then displaced and an ether is formed. This reaction mechanism does not favour the formation of bulky ethers. Hence, if the alkyl halide is secondary or tertiary, the reaction that happens is an elimination reaction instead of a displacement reaction due to steric hindrance.
The elimination occurs by the ${{E}_{1}}cB$ mechanism of elimination and dehydrohalogenation takes place. Thus, an alkene is formed. The reaction that occurs will be:

Thus, as we can see, dehydrohalogenation occurs on the tertiary carbon and an adjacent carbon. A double bond forms between them and formation of an alkene occurs.
Hence, the answer is ‘B. isobutylene’
Note: Please do not get confused due to the fact that this is a Williamson synthesis. Remember that only primary alkyl halides form ethers. Because of this, do not blindly mark your answer as ‘C. t-butyl methyl ether’. Always check if the alkyl halide is primary, secondary, or tertiary before working out the reaction and marking the answer.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




