The descending order of stability of the following carbonium ions is?
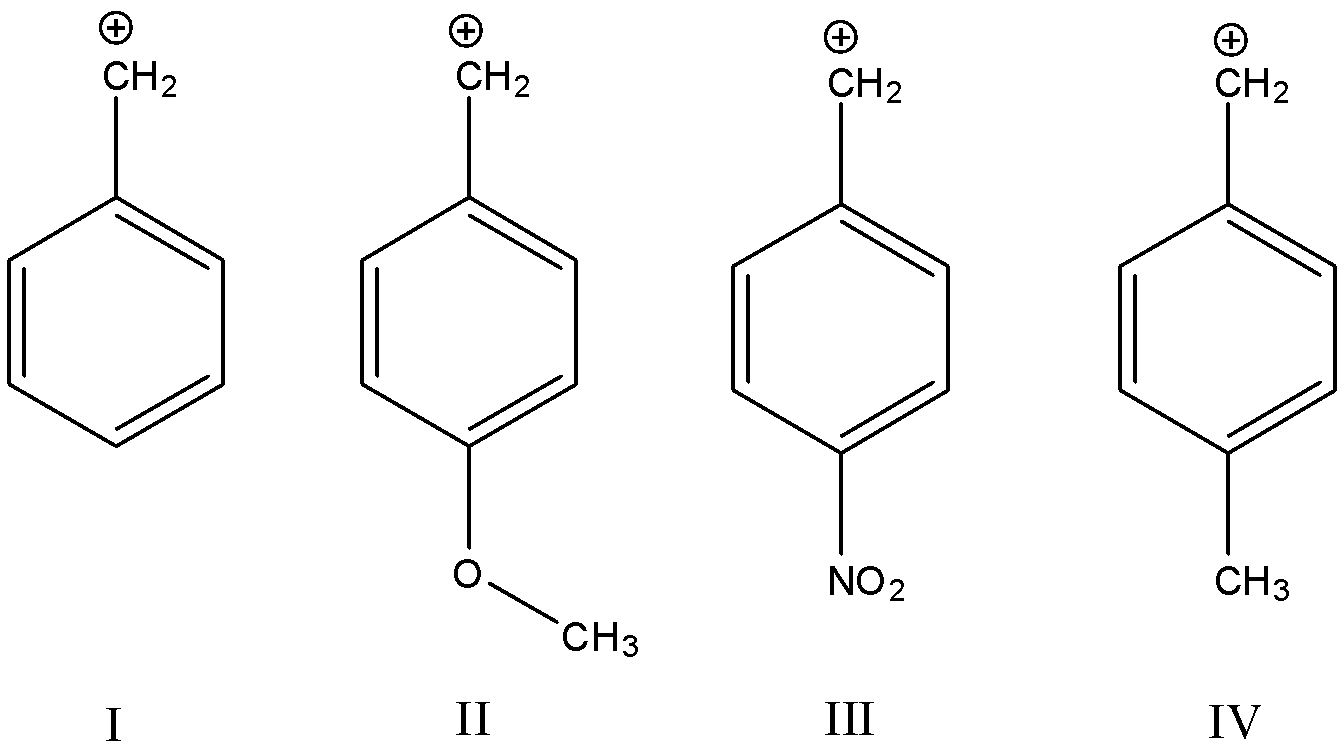
A. IV > II > I > III
B. II > IV > III > I
C. II > IV > I > III
D. IV > II > III > I
Answer
267.3k+ views
Hint: Think about how electron-donating or electron-withdrawing groups affect the stability of the carbonium ion. Classify the given compounds with respect to the functional groups that are attached opposite to the carbonium carbon ion as electron-donating or electron-withdrawing groups.
Complete step by step solution:
We know that the stability of the carbonium ion increases with the presence of electron-donating groups and decreases with the presence of electron-withdrawing groups. The increase or decrease instability is considered with respect to the base molecule that does not have an electron-withdrawing or donating groups attached to it. Let us consider the given structures one by one to determine which one is more stable.
-I: In this molecule, there are no electron-donating or withdrawing groups present, so we will consider this as the base molecule and check the stability of the other molecules with respect to this.
-II: In this molecule, a methoxy group ($-OC{{H}_{3}}$) is present in the para position with respect to the carbonium ion. We know that the methoxy group is a slightly electron-donating group. The inductive effect of the methyl group in the methoxy group gives it this character. So, this molecule will be more stable than our base molecule I.
-III: In this molecule, a nitro group ($-N{{O}_{2}}$) is present in the para position with respect to the carbonium ion. We know that the nitro group is a strongly electron-withdrawing group and will decrease the stability of the carbonium ion. So, this molecule will be less table than our base molecule I.
-IV: In this molecule, a methyl group ($-C{{H}_{3}}$) is present in the para position with respect to the carbonium ion. We know that the methyl group is a very good electron-donating group and will increase the stability of the carbonium ion. So, this molecule will be more stable than our base molecule I as well as molecule II.
Hence, the correct order of decreasing stability of the carbonium ion is A. “IV > II > I > III”
Note: Remember that although the presence of the oxygen atom in the methoxy group would indicate that it may be an electron-withdrawing group, that is not the case due to the presence of the methyl group and its inductive effect. The longer the alkyl chain in the alkoxy group, the greater will be the tendency to donate electrons.
Complete step by step solution:
We know that the stability of the carbonium ion increases with the presence of electron-donating groups and decreases with the presence of electron-withdrawing groups. The increase or decrease instability is considered with respect to the base molecule that does not have an electron-withdrawing or donating groups attached to it. Let us consider the given structures one by one to determine which one is more stable.
-I: In this molecule, there are no electron-donating or withdrawing groups present, so we will consider this as the base molecule and check the stability of the other molecules with respect to this.
-II: In this molecule, a methoxy group ($-OC{{H}_{3}}$) is present in the para position with respect to the carbonium ion. We know that the methoxy group is a slightly electron-donating group. The inductive effect of the methyl group in the methoxy group gives it this character. So, this molecule will be more stable than our base molecule I.
-III: In this molecule, a nitro group ($-N{{O}_{2}}$) is present in the para position with respect to the carbonium ion. We know that the nitro group is a strongly electron-withdrawing group and will decrease the stability of the carbonium ion. So, this molecule will be less table than our base molecule I.
-IV: In this molecule, a methyl group ($-C{{H}_{3}}$) is present in the para position with respect to the carbonium ion. We know that the methyl group is a very good electron-donating group and will increase the stability of the carbonium ion. So, this molecule will be more stable than our base molecule I as well as molecule II.
Hence, the correct order of decreasing stability of the carbonium ion is A. “IV > II > I > III”
Note: Remember that although the presence of the oxygen atom in the methoxy group would indicate that it may be an electron-withdrawing group, that is not the case due to the presence of the methyl group and its inductive effect. The longer the alkyl chain in the alkoxy group, the greater will be the tendency to donate electrons.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




