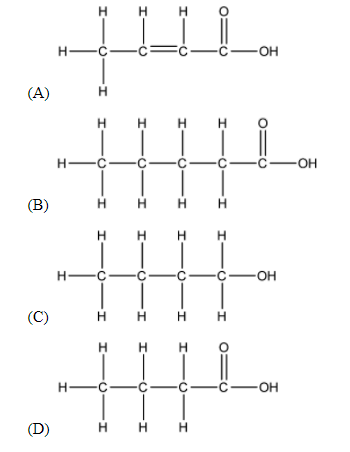
The correct structural formula of butanoic acid is:
Answer
268.8k+ views
Hint: The term ‘-oic acid’ suffix suggests the presence of a carboxylic acid functional group in the organic compound but prefix suggests the presence of four carbons in the compound.
Complete step by step answer:
Let’s get familiar about nomenclature.In general, an IUPAC name always has three essential features:
i) A root or base indicating a major chain or ring of carbon atoms found in the molecular structure.
ii) A suffix or other element(s) which represents the functional groups that may be present in the compound.
iii) Names of substituent groups, other than hydrogen, that complete the molecular structure.
With these rules in mind, let us now analyse the name ‘butanoic acid’.
The prefix ‘but-’ means that the longest Carbon chain of the required structure contains 4 Carbon atoms. Thus, we can easily eliminate (B) as a possible answer.
As the suffix ‘-oic acid’ suggests, the functional group of the compound is carboxylic acid. Carboxylic acid can be shown as –COOH. This further narrows our possible answers down to (A) and (D).
The middle part of the name ‘-an-’ suggests that there are no unsaturated bonds in the longest Carbon chain, thus we can eliminate option (A) as an answer. Thus, we can safely conclude that the answer to this question is (D).
Thus, the answer to the above question is option (D).
Note: Here, we are given only ‘butanoic acid’ as the name of the compound, so terminal carbon will be of carboxylic acid functional group. When any specific number is given before the name of the compound (i.e. 2-butanoic acid), then it will suggest the position of the carboxylic acid functional group and we will then have to choose the structure accordingly.
Complete step by step answer:
Let’s get familiar about nomenclature.In general, an IUPAC name always has three essential features:
i) A root or base indicating a major chain or ring of carbon atoms found in the molecular structure.
ii) A suffix or other element(s) which represents the functional groups that may be present in the compound.
iii) Names of substituent groups, other than hydrogen, that complete the molecular structure.
With these rules in mind, let us now analyse the name ‘butanoic acid’.
The prefix ‘but-’ means that the longest Carbon chain of the required structure contains 4 Carbon atoms. Thus, we can easily eliminate (B) as a possible answer.
As the suffix ‘-oic acid’ suggests, the functional group of the compound is carboxylic acid. Carboxylic acid can be shown as –COOH. This further narrows our possible answers down to (A) and (D).
The middle part of the name ‘-an-’ suggests that there are no unsaturated bonds in the longest Carbon chain, thus we can eliminate option (A) as an answer. Thus, we can safely conclude that the answer to this question is (D).
Thus, the answer to the above question is option (D).
Note: Here, we are given only ‘butanoic acid’ as the name of the compound, so terminal carbon will be of carboxylic acid functional group. When any specific number is given before the name of the compound (i.e. 2-butanoic acid), then it will suggest the position of the carboxylic acid functional group and we will then have to choose the structure accordingly.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




