The compound used in the manufacture of terylene is
A. Ethylene
B. Vinyl chloride
C. Ethylene glycol
D. Adipic acid
Answer
249.9k+ views
Hint: Terylene is a synthetic polyester fibre based on terephthalic acid. It is light in weight and it is used in clothing, sheets, ropes.
Complete Step by Step Solution:
Terylene is manufactured by the condensation of ethylene glycol and Terephthalic acid (1,4-benzene dicarboxylic acid) with removal of water in this zinc acetate and antimony trioxide mixture is used as catalyst. The temperature required for this process is 151.85-degree Celsius to 201.85-degree Celsius. In polymerization many monomers react together and form polymers. Terylene is also known as Dacron.
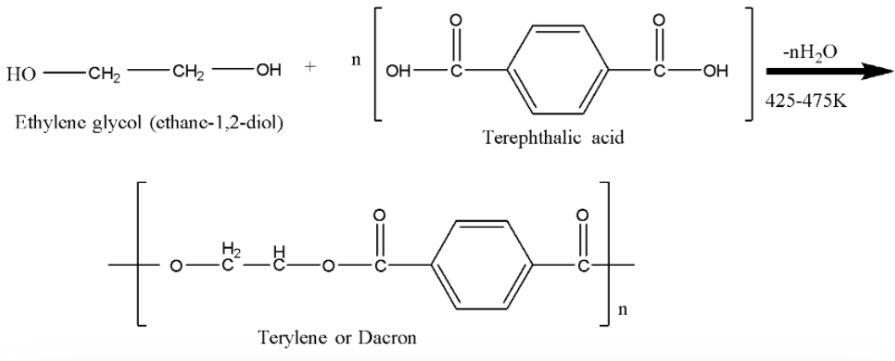
Here we can see that there are two monomers of ethylene glycol and terephthalic acid (1,4-benzene dicarboxylic acid) which are condensed and after the removal of water we get Terylene or Dacron. In this one linkage is present called ester linkage. Ester linkage is given below
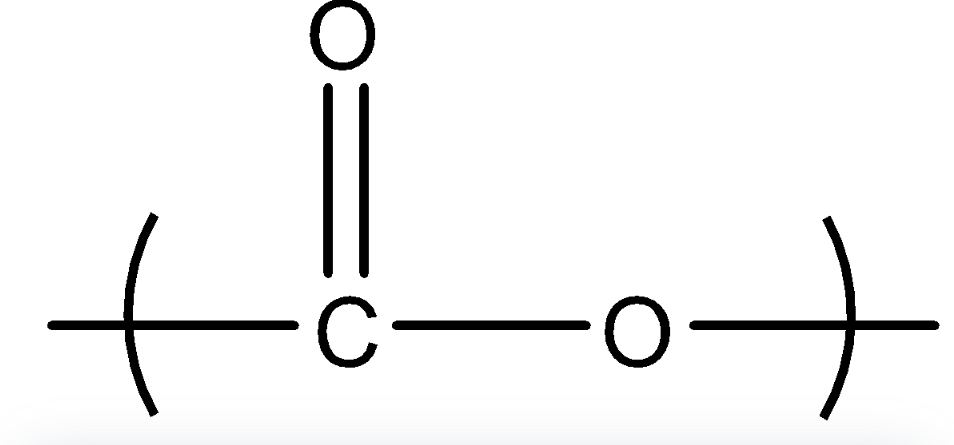
Image: Ester linkage
So, correct answer of the question is option (C) “Ethylene glycol”
Additional information:
Ethylene glycol and terephthalic acid is obtained from petroleum. This fibre was first created in 1941 by chemist J R Whinfield of Carington. The major disadvantage is they are not easily procurable by the yard. Terylene is majorly used in the textile industry to make many dresses. It is easily washed and dried quickly.it is used for laundry usage as an automatic clothing vacuum packing machine and also used for polyester tricot knit.
Note: Transesterification followed by polymerization gives terylene. Terylene is the strongest polymer. It is used to make non-woven needle punched carpet for exhibition.
Complete Step by Step Solution:
Terylene is manufactured by the condensation of ethylene glycol and Terephthalic acid (1,4-benzene dicarboxylic acid) with removal of water in this zinc acetate and antimony trioxide mixture is used as catalyst. The temperature required for this process is 151.85-degree Celsius to 201.85-degree Celsius. In polymerization many monomers react together and form polymers. Terylene is also known as Dacron.

Here we can see that there are two monomers of ethylene glycol and terephthalic acid (1,4-benzene dicarboxylic acid) which are condensed and after the removal of water we get Terylene or Dacron. In this one linkage is present called ester linkage. Ester linkage is given below

Image: Ester linkage
So, correct answer of the question is option (C) “Ethylene glycol”
Additional information:
Ethylene glycol and terephthalic acid is obtained from petroleum. This fibre was first created in 1941 by chemist J R Whinfield of Carington. The major disadvantage is they are not easily procurable by the yard. Terylene is majorly used in the textile industry to make many dresses. It is easily washed and dried quickly.it is used for laundry usage as an automatic clothing vacuum packing machine and also used for polyester tricot knit.
Note: Transesterification followed by polymerization gives terylene. Terylene is the strongest polymer. It is used to make non-woven needle punched carpet for exhibition.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




