The chemical formula of iron (iii) carbonate is:
A. $F{e_2}{\left( {C{O_3}} \right)_2}$
B. $F{e_2}\left( {C{O_3}} \right)$
C. $F{e_2}{\left( {C{O_3}} \right)_3}$
D. $Fe{\left( {C{O_3}} \right)_2}$
Answer
268.8k+ views
Hint: Every chemical compound has a fixed composition and number of atoms present in the compound. Chemical formula is a set of chemical symbols showing the elements present in a compound and their relative proportions.
Complete step by step answer:
A chemical formula tells us the number of atoms of each element in a compound. It contains symbols of the atoms of the elements present in the compound. Each chemical substance has a specific chemical composition.
Steps for writing a chemical formula:
(i) First, you have to decide the type of bonds either covalent bond or ionic bond.
(ii) Write the symbol of a polyatomic ion or the element.
(iii) Write the valency of each atom or group of atoms.
(iv) Now, after writing the valency of each atom or group of atoms, balance the charge of each species.
Given:
Chemical formula of iron (iii) carbonate is
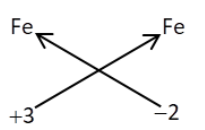
The chemical formula of iron (iii) carbonate is $F{e_2}{\left( {C{O_3}} \right)_2}$
Hence, the correct option is (C).
Note:
Monoatomic ions are composed of single atoms and polyatomic ions are composed of two or more atoms per ion.
Complete step by step answer:
A chemical formula tells us the number of atoms of each element in a compound. It contains symbols of the atoms of the elements present in the compound. Each chemical substance has a specific chemical composition.
Steps for writing a chemical formula:
(i) First, you have to decide the type of bonds either covalent bond or ionic bond.
(ii) Write the symbol of a polyatomic ion or the element.
(iii) Write the valency of each atom or group of atoms.
(iv) Now, after writing the valency of each atom or group of atoms, balance the charge of each species.
Given:
Chemical formula of iron (iii) carbonate is
| Symbol of atomsOr group of atoms | $Fe$ | $C{O_3}$ |
| Charge | $ + 3$ | $ - 2$ |

The chemical formula of iron (iii) carbonate is $F{e_2}{\left( {C{O_3}} \right)_2}$
Hence, the correct option is (C).
Note:
Monoatomic ions are composed of single atoms and polyatomic ions are composed of two or more atoms per ion.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




