The carbyl amine reaction is:
(A) 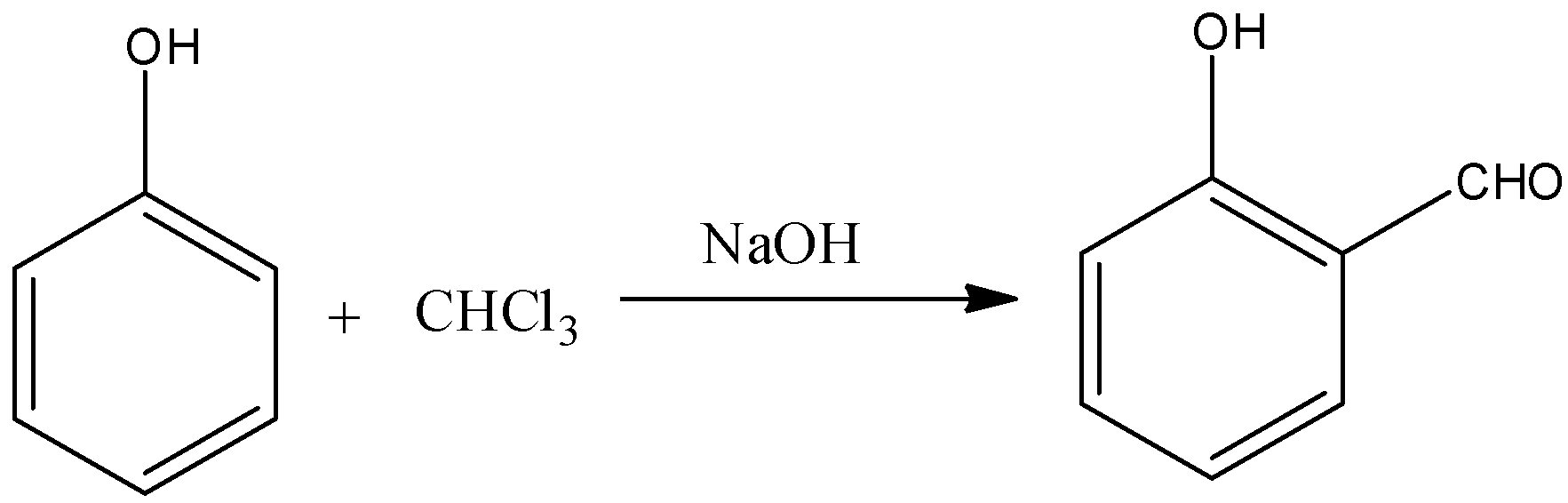
(B) 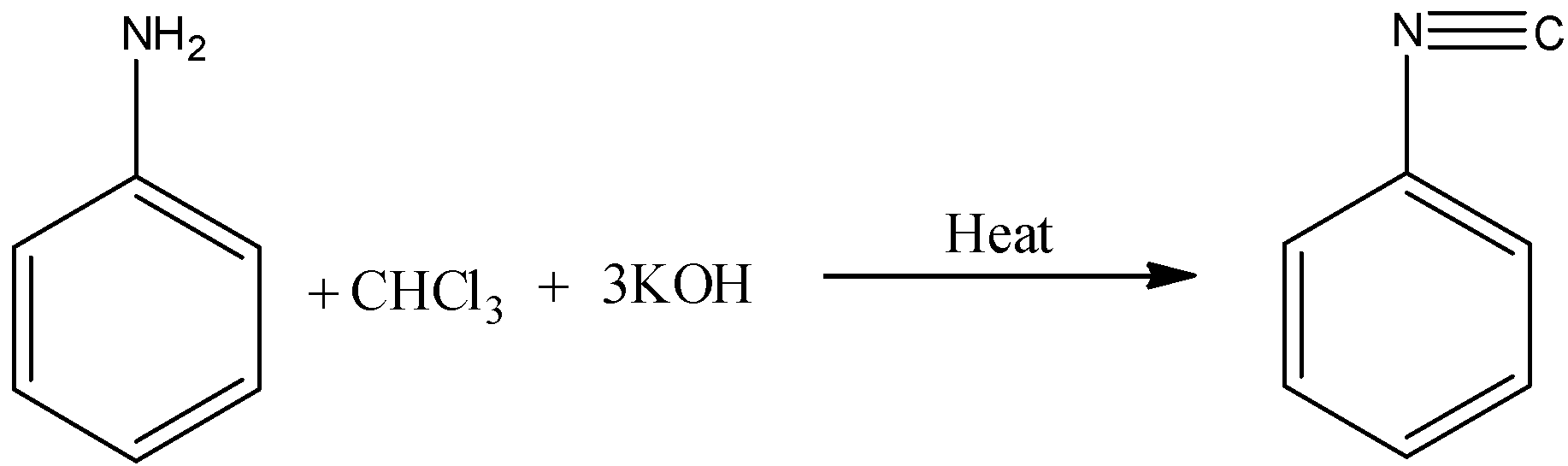
(C) 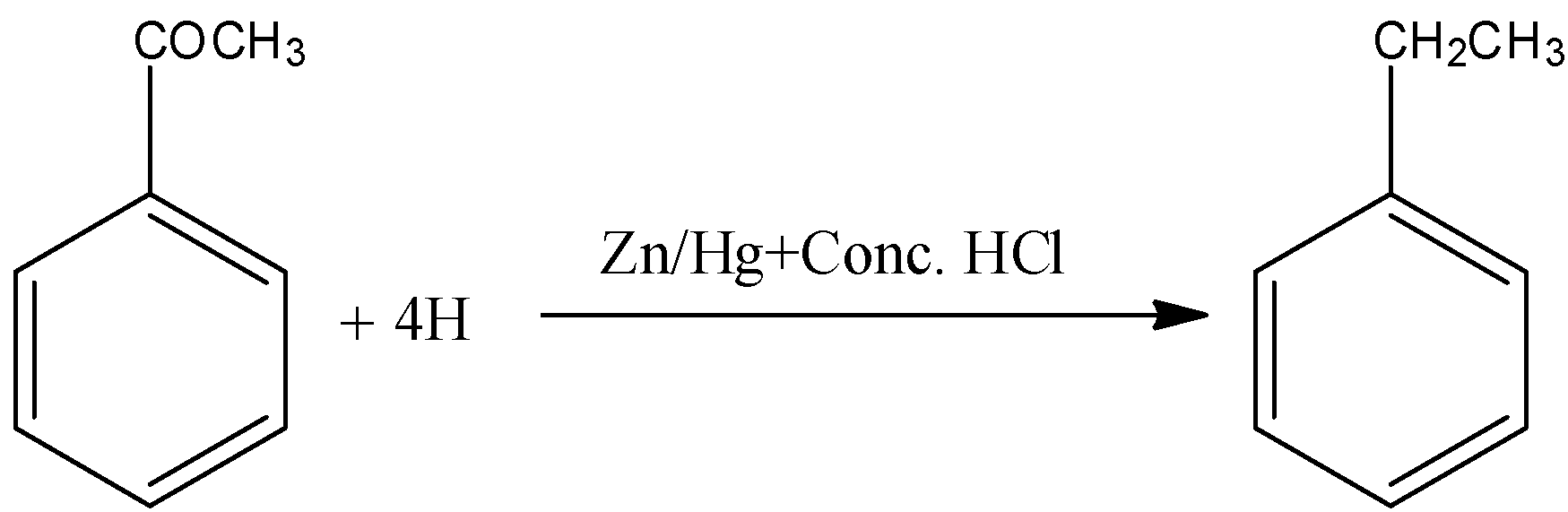
(D) 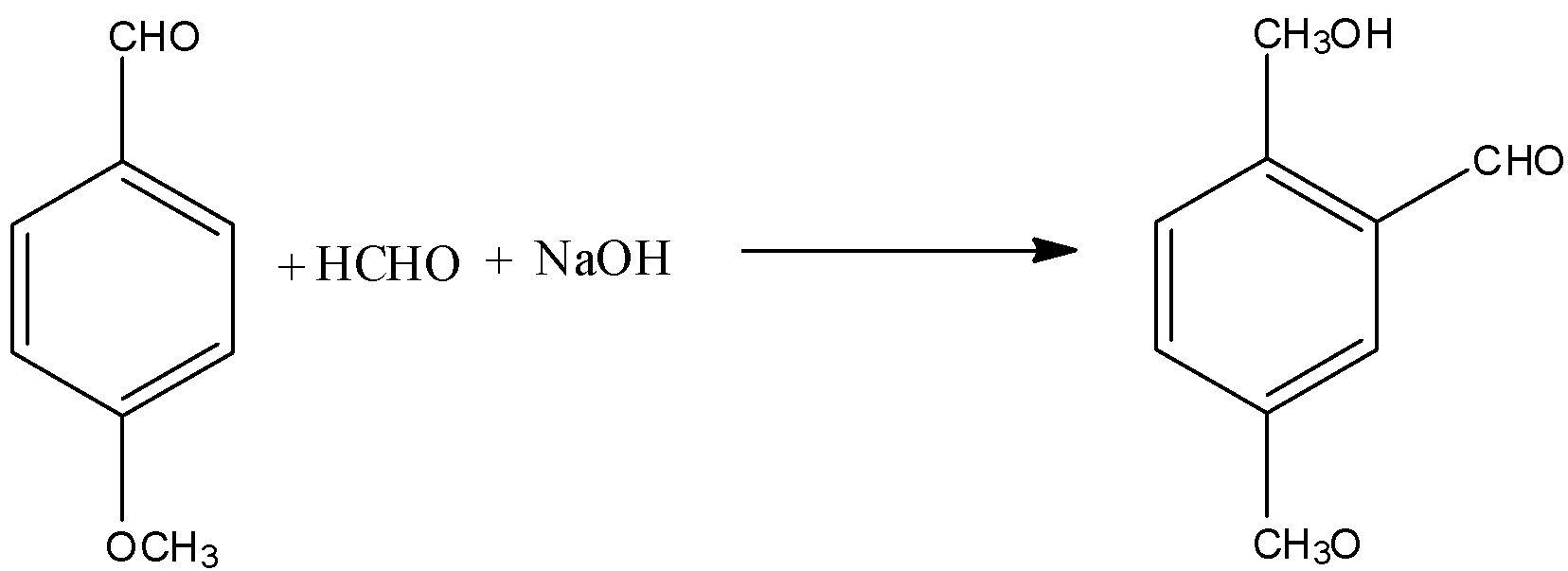
Answer
264.6k+ views
Hint: The carbylamine reaction is also known as the Hofmann isocyanide synthesis. It is used to test the presence of primary amines. If primary amines are present then they convert into carbonyl amines which will give a foul smell.
Complete step by step answer:
-Carbylamine is also called Isocyanide or Isonitrile. Carbylamines are a class of organic compounds having the molecular structure\[R-\overset{+}{\mathop{N}}\,\equiv C\] here R = organic compound.
-Primary amines on warming with chloroform and alcoholic KOH gives carbylamine (Isocyanide) as the product.
-In this reaction carbylamine is going to form as a product then this reaction is called carbylamine reaction.
-The reaction is as follows.
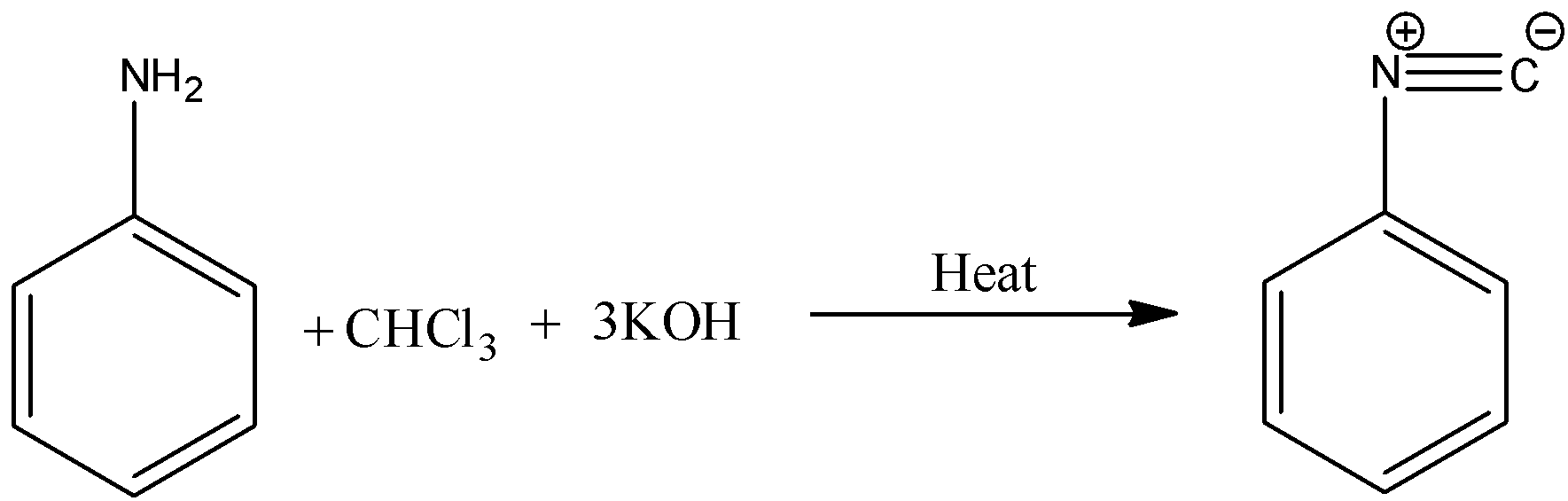
-The reaction mechanism of carbylamine reaction is as follows.
-The reaction mechanism includes the reaction of a primary amine to the intermediate (dichloroCarbene) formed from the dehydrohalogenation of chloroform. This intermediate is called dichlorocarbene
-The heat in the reaction is required to generate the dichlorocarbene from chloroform.
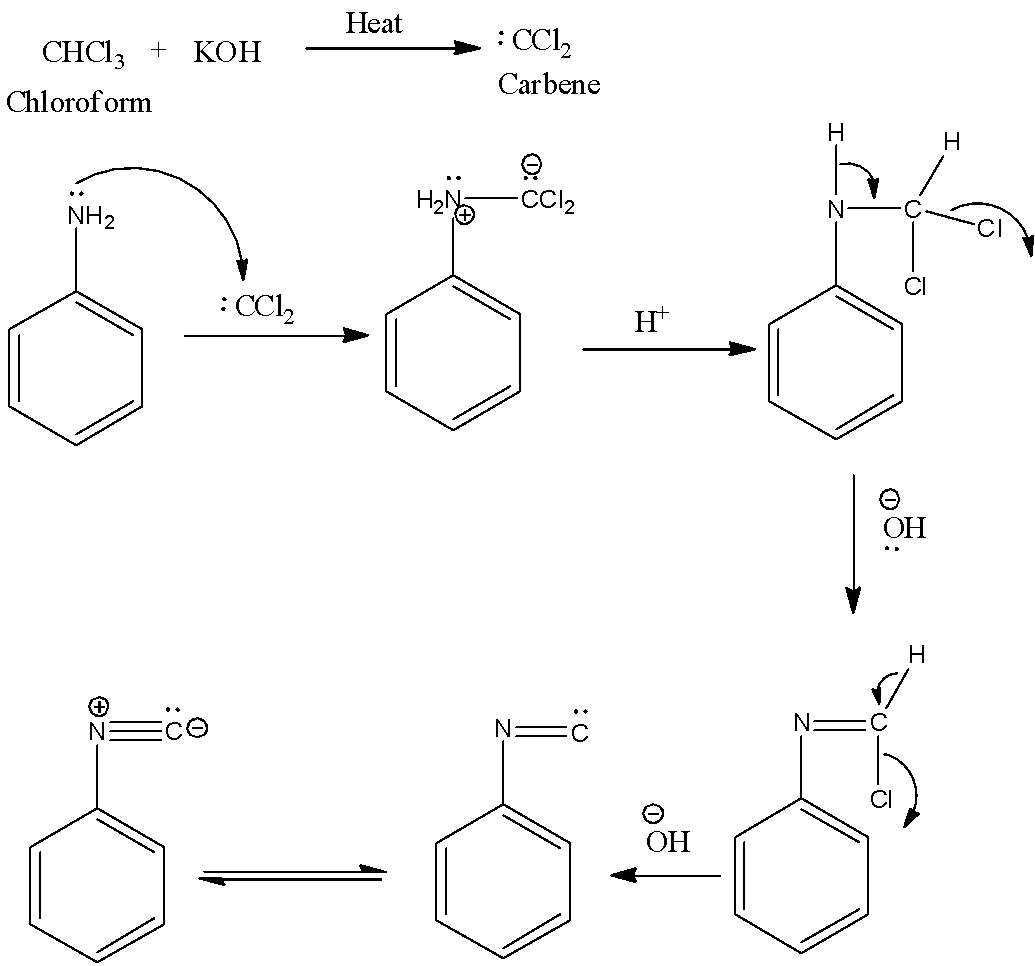
-Therefore the primary amines are going to react with chloroform in presence of alcoholic KOH gives isocyanide as the product.
So, the correct option is B.
Additional information:
-Secondary amines and tertiary amines do not respond to carbylamine tests.
-Both aliphatic and aromatic primary amines are going to respond with carbylamine reaction.
Note: The formation of isocyanide can be easily identified with strong foul smell. Strong foul smell is an indication for the presence of primary amines in the given compounds. This reaction is introduced by Hoffman, so this reaction is also called Hoffman isocyanide synthesis.
Complete step by step answer:
-Carbylamine is also called Isocyanide or Isonitrile. Carbylamines are a class of organic compounds having the molecular structure\[R-\overset{+}{\mathop{N}}\,\equiv C\] here R = organic compound.
-Primary amines on warming with chloroform and alcoholic KOH gives carbylamine (Isocyanide) as the product.
-In this reaction carbylamine is going to form as a product then this reaction is called carbylamine reaction.
-The reaction is as follows.

-The reaction mechanism of carbylamine reaction is as follows.
-The reaction mechanism includes the reaction of a primary amine to the intermediate (dichloroCarbene) formed from the dehydrohalogenation of chloroform. This intermediate is called dichlorocarbene
-The heat in the reaction is required to generate the dichlorocarbene from chloroform.

-Therefore the primary amines are going to react with chloroform in presence of alcoholic KOH gives isocyanide as the product.
So, the correct option is B.
Additional information:
-Secondary amines and tertiary amines do not respond to carbylamine tests.
-Both aliphatic and aromatic primary amines are going to respond with carbylamine reaction.
Note: The formation of isocyanide can be easily identified with strong foul smell. Strong foul smell is an indication for the presence of primary amines in the given compounds. This reaction is introduced by Hoffman, so this reaction is also called Hoffman isocyanide synthesis.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




