Nitrene is an intermediate in one of the following reactions:
A. Schmidt rearrangement
B. Beckmann rearrangement
C. Baeyer-Villiger oxidation
D. Curtius reaction
Answer
264k+ views
Hint : Nitrene is the nitrogen analogue of a carbene. It is an electrophile because it has five valence electrons. It is involved in many chemical reactions and acts as a reactive intermediate. It can be generated by two common ways. It can be generated by thermolysis or photolysis of azides with expulsion of nitrogen gas. Another way of generation of nitrene is the expulsion of carbon monoxide.
Complete step by step solution:
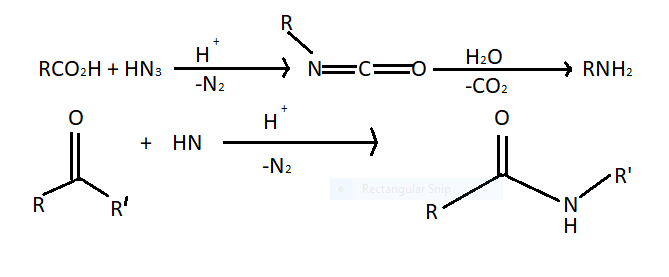
In Schmidt rearrangement alkyl migration over carbon nitrogen bonds takes place. This reaction refers to a reaction between an acid catalyst hydrazoic acid with electrophiles. Carboxylic acid forms amines through an isocyanate intermediate and ketones from amides. Nitrene is an intermediate in the following reactions.
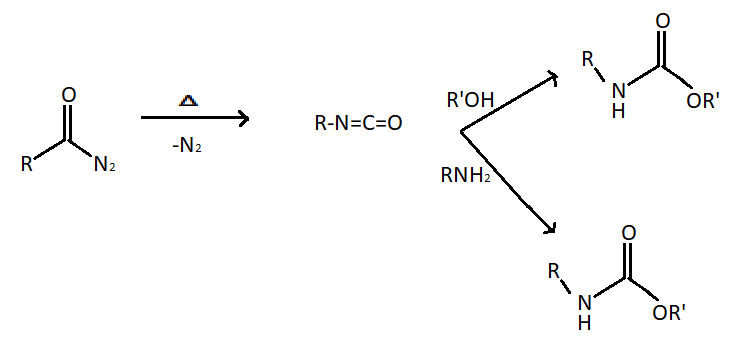
In the Curtius reaction, the production of an isocyanate is accomplished by the thermal decomposition of carboxylic azides. The mechanism involves the alkyl shift of the $R$ group from carbonyl carbon to the closet nitrogen. The reaction has similarities with Schmidt reaction with acids but differs in that the acyl azide in the present case is prepared from acyl halide and an azide salt.
Hence option A and D both are correct answers of this problem because in both of the reactions nitrene is an intermediate.
Note : Nitrenes are highly reactive molecule species with a monovalent nitrogen atom which can exist in a singlet and triplet state. Nitrenes are more stable than carbene. It is because of greater thermodynamic stability of nitrene. As nitrenes are very reactive they are not isolated. The insertion of nitrene can be easily into a carbon to hydrogen covalent bond yielding an amine or amide.
Complete step by step solution:
In Schmidt rearrangement alkyl migration over carbon nitrogen bonds takes place. This reaction refers to a reaction between an acid catalyst hydrazoic acid with electrophiles. Carboxylic acid forms amines through an isocyanate intermediate and ketones from amides. Nitrene is an intermediate in the following reactions.

In the Curtius reaction, the production of an isocyanate is accomplished by the thermal decomposition of carboxylic azides. The mechanism involves the alkyl shift of the $R$ group from carbonyl carbon to the closet nitrogen. The reaction has similarities with Schmidt reaction with acids but differs in that the acyl azide in the present case is prepared from acyl halide and an azide salt.

Hence option A and D both are correct answers of this problem because in both of the reactions nitrene is an intermediate.
Note : Nitrenes are highly reactive molecule species with a monovalent nitrogen atom which can exist in a singlet and triplet state. Nitrenes are more stable than carbene. It is because of greater thermodynamic stability of nitrene. As nitrenes are very reactive they are not isolated. The insertion of nitrene can be easily into a carbon to hydrogen covalent bond yielding an amine or amide.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




