The basicity of phosphoric acid is:-
(a)- 1
(b)- 2
(c)- 3
(d)- 4
Answer
259.8k+ views
Hint: For checking the basicity of a compound the structure is drawn. The type of bonds the central metal atom forms and the group joined to the metal atom is to be found out. Electronegativity of the atom is also checked.
Complete step by step answer:
When oxoacids of phosphorus are formed, it shows basic character.
This basic character is because of the presence of an OH group present in the compound.
The OH group present is ionizable because the H atom is attached to a highly electronegative atom O (oxygen). When dissolved in water the hydrogen ions get separated which tells the basicity of the compound.
So, to check the basicity of the compound we have to count the number of OH group present which are directly attached to the central atom
Phosphoric acid has a formula \[{{H}_{3}}P{{O}_{4}}\].
In this, the phosphorus has a +5 oxidation state.
Let us now see the structure of the phosphoric acid \[{{H}_{3}}P{{O}_{4}}\].
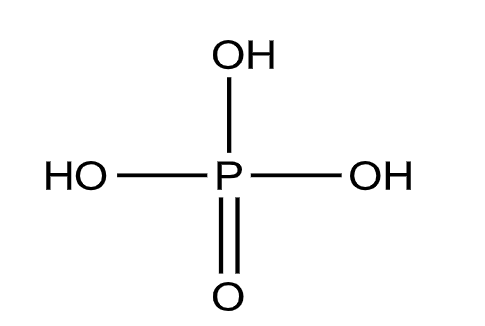
From the structure, we can see that there are three P-OH bonds and one P=O bond.
So, due to the presence of three P-OH bonds, it can give 3 hydrogen ions when dissolved in water.
Hence the correct option is (c)- 3.
Additional information:
The basicity of some oxoacids of phosphorus are:
The phosphinic acid (\[{{H}_{3}}P{{O}_{2}}\]) has basicity 1
The hypophosphoric acid (\[{{H}_{4}}{{P}_{2}}{{O}_{6}}\]) has basicity 4.
The peroxodiphosphoric acid (\[{{H}_{4}}{{P}_{2}}{{O}_{8}}\]) has basicity 4.
Note:
The chemical properties of the members of a homologous series similar though the first member may vary considerably from the rest of the members. The successive members of a homologous series differ by a \[C{{H}_{2}}\] group or by 14 mass units.
Complete step by step answer:
When oxoacids of phosphorus are formed, it shows basic character.
This basic character is because of the presence of an OH group present in the compound.
The OH group present is ionizable because the H atom is attached to a highly electronegative atom O (oxygen). When dissolved in water the hydrogen ions get separated which tells the basicity of the compound.
So, to check the basicity of the compound we have to count the number of OH group present which are directly attached to the central atom
Phosphoric acid has a formula \[{{H}_{3}}P{{O}_{4}}\].
In this, the phosphorus has a +5 oxidation state.
Let us now see the structure of the phosphoric acid \[{{H}_{3}}P{{O}_{4}}\].

From the structure, we can see that there are three P-OH bonds and one P=O bond.
So, due to the presence of three P-OH bonds, it can give 3 hydrogen ions when dissolved in water.
Hence the correct option is (c)- 3.
Additional information:
The basicity of some oxoacids of phosphorus are:
The phosphinic acid (\[{{H}_{3}}P{{O}_{2}}\]) has basicity 1
The hypophosphoric acid (\[{{H}_{4}}{{P}_{2}}{{O}_{6}}\]) has basicity 4.
The peroxodiphosphoric acid (\[{{H}_{4}}{{P}_{2}}{{O}_{8}}\]) has basicity 4.
Note:
The chemical properties of the members of a homologous series similar though the first member may vary considerably from the rest of the members. The successive members of a homologous series differ by a \[C{{H}_{2}}\] group or by 14 mass units.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




