Reagent, 1-naphthylamine, and sulfanilic acid in acetic acid are used for the detection of
A. \[{\rm{NO}}\]
B. \[{{\rm{N}}_{\rm{2}}}{\rm{O}}\]
C. \[{\rm{N}}{{\rm{O}}^{\rm{3}}}^{\rm{ - }}\]
D. \[{\rm{N}}{{\rm{O}}^{\rm{2}}}^{\rm{ - }}\]
Answer
266.7k+ views
Hint: When an aromatic amine reacts with nitrous acid at 273-278K forms arene diazonium salt. This arene diazonium salt on treatment with amines forms brightly coloured azo dye.
Complete Step by Step Solution:
Nitrite ions combine with acetic acid to form nitrous acid and acetate ions.
\[{\rm{N}}{{\rm{O}}^{{\rm{2 - }}}}{\rm{ + C}}{{\rm{H}}_{\rm{3}}}{\rm{COOH}} \to {\rm{HN}}{{\rm{O}}_2}{\rm{ + C}}{{\rm{H}}_{\rm{3}}}{\rm{CO}}{{\rm{O}}^{\rm{ - }}}\]
This nitrous acid then reacts with sulfanilic acid.
When sulfanilic acid is added to a 1-naphthylamine reagent acidified with acetic acid, sulphanilic acid undergoes diazotization in presence of nitrous acid.
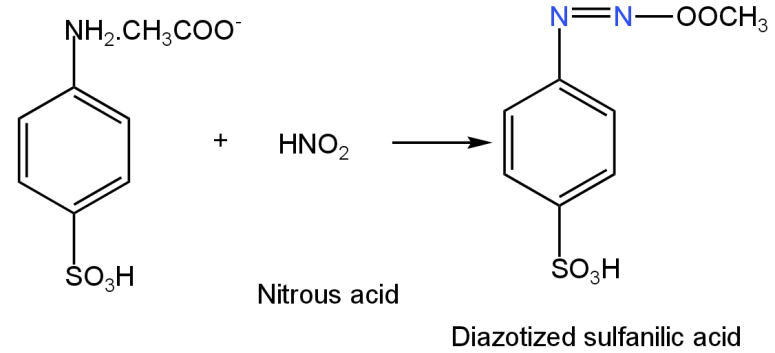
Image: Formation of diazotized sulfanilic acid.
Diazotized sulfanilic acid when treated with 1-naphthyl amine undergo coupling reaction.
Red azo dye is formed as a product. The IUPAC name of the compound is p-sulfobenzene-azo-1-naphthyl amine.
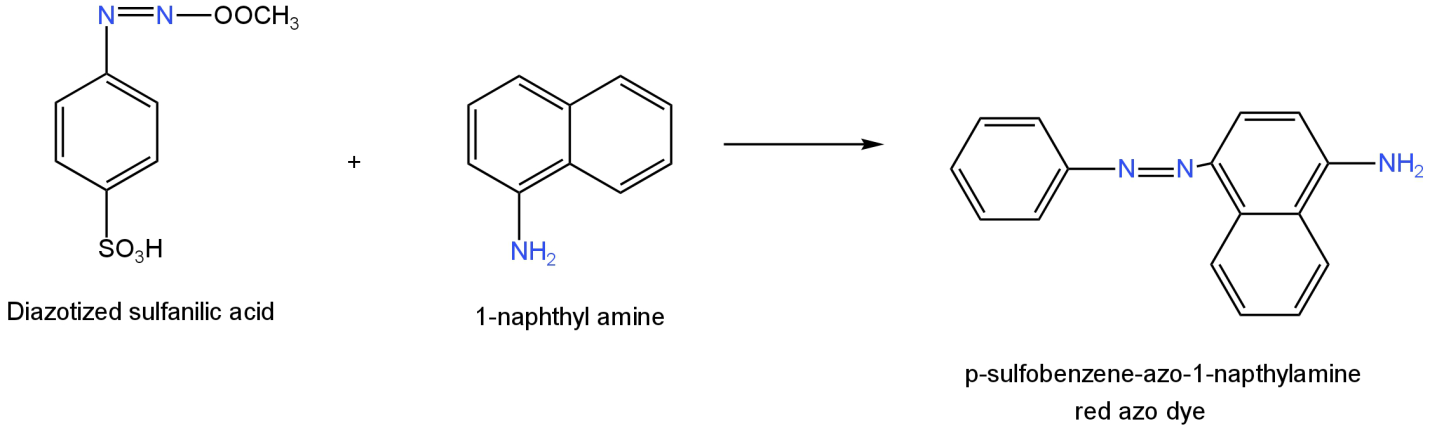
Image: Reaction of diazotized sulfanilic acid with 1-naphthyl amine.
This confirms the presence of nitrite ions in a given solution.
Sulphanilic acid and 1-naphthylamine reagent test or the Griess-Ilosvay test is the test for nitrite ions.
So, option D is correct.
Additional information: The well-known indicator methyl orange which is widely utilized in acid-base titrations is obtained by coupling the diazonium salt of sulfanilic acid with N, N-dimethylaniline.
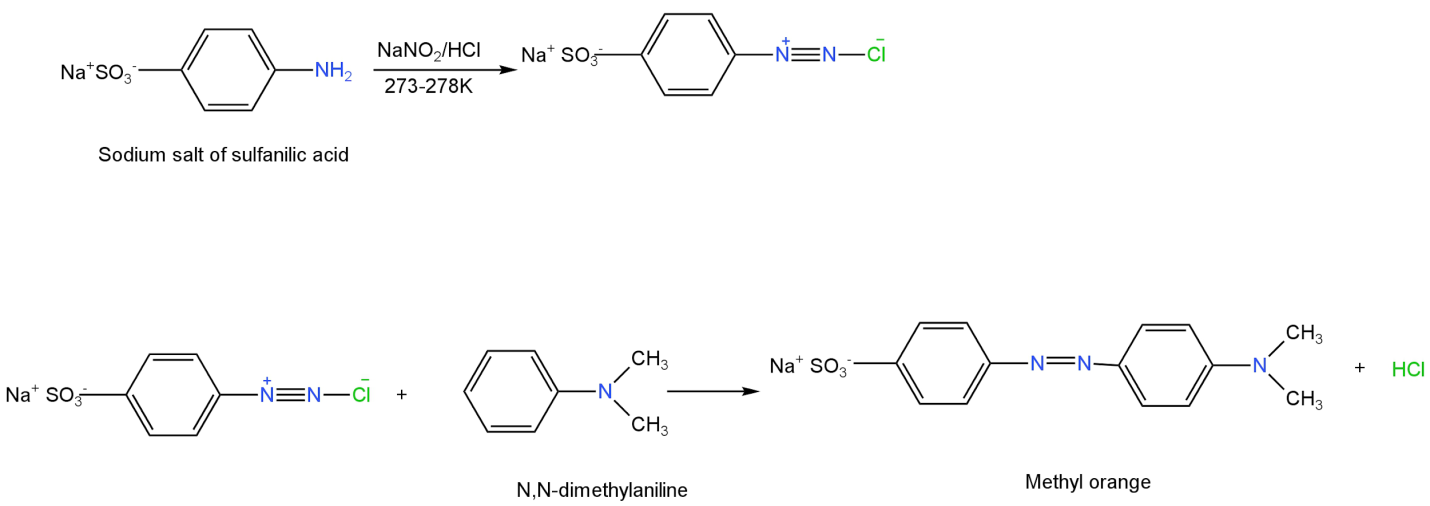
Image: Formation of methyl orange.
Note: The coupling reaction of the arene diazonium salt with amine happens in a faintly acidic medium(pH 4-5) at 273-278K. The coupling reaction is an example of an electrophilic substitution reaction.In this reaction, the diazonium action with the positive charge on the terminal nitrogen acts as the electrophile. The electron-rich compound amine acts as the nucleophile. Coupling occurs at the para position with the amino group. The colour of the azo compounds is due to the extended conjugation involving the double bonds of both the arene (benzene) rings through the \[{\rm{ - N = N - }}\] double bond.
Complete Step by Step Solution:
Nitrite ions combine with acetic acid to form nitrous acid and acetate ions.
\[{\rm{N}}{{\rm{O}}^{{\rm{2 - }}}}{\rm{ + C}}{{\rm{H}}_{\rm{3}}}{\rm{COOH}} \to {\rm{HN}}{{\rm{O}}_2}{\rm{ + C}}{{\rm{H}}_{\rm{3}}}{\rm{CO}}{{\rm{O}}^{\rm{ - }}}\]
This nitrous acid then reacts with sulfanilic acid.
When sulfanilic acid is added to a 1-naphthylamine reagent acidified with acetic acid, sulphanilic acid undergoes diazotization in presence of nitrous acid.

Image: Formation of diazotized sulfanilic acid.
Diazotized sulfanilic acid when treated with 1-naphthyl amine undergo coupling reaction.
Red azo dye is formed as a product. The IUPAC name of the compound is p-sulfobenzene-azo-1-naphthyl amine.

Image: Reaction of diazotized sulfanilic acid with 1-naphthyl amine.
This confirms the presence of nitrite ions in a given solution.
Sulphanilic acid and 1-naphthylamine reagent test or the Griess-Ilosvay test is the test for nitrite ions.
So, option D is correct.
Additional information: The well-known indicator methyl orange which is widely utilized in acid-base titrations is obtained by coupling the diazonium salt of sulfanilic acid with N, N-dimethylaniline.

Image: Formation of methyl orange.
Note: The coupling reaction of the arene diazonium salt with amine happens in a faintly acidic medium(pH 4-5) at 273-278K. The coupling reaction is an example of an electrophilic substitution reaction.In this reaction, the diazonium action with the positive charge on the terminal nitrogen acts as the electrophile. The electron-rich compound amine acts as the nucleophile. Coupling occurs at the para position with the amino group. The colour of the azo compounds is due to the extended conjugation involving the double bonds of both the arene (benzene) rings through the \[{\rm{ - N = N - }}\] double bond.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




