Primary nitro compounds when react with \[HN{{O}_{2}}\] from crystalline solids which on reaction with \[NaOH~\] gives
(A) Red solution
(B) Blue solution
(C) White precipitate
(D) Yellow coloration
Answer
268.5k+ views
Hint: Primary nitro compound is one in which the\[N{{O}_{2}}\] group is attached to one carbon which is further bonded with one alkyl chain or an alkyl group such as\[RC{{H}_{2}}N{{O}_{2}}\]. The reaction of this primary nitro compound with nitrous (\[HN{{O}_{2}}\]) results in releases of water molecules to give nitrolic acid. Again nitrolic acid reacts with \[NaOH~\](caustic soda) to release water molecules to give a soluble sodium salt.
Complete Step by Step Solution:
Now let us take a primary nitro alkyl compound, nitro ethane (such as\[C{{H}_{3}}C{{H}_{2}}N{{O}_{2}}\]which on reacting with nitrous reagent (\[HN{{O}_{2}}\]) gives nitrolic acid (double bond between carbon and nitrogen). In this, two hydrogen of first carbon from the nitro group makes the bond with one oxygen of nitrous to release water molecule such as
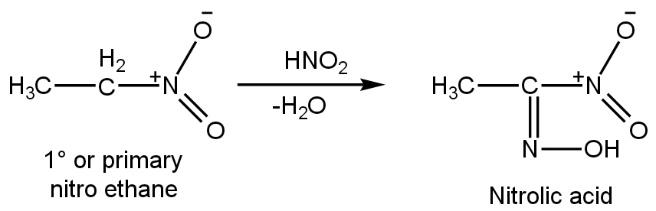
Now this nitrolic acid reacts with \[NaOH~\](caustic soda) to give soluble sodium salt because after reacting nitrolic acid with \[NaOH~\], the water molecule (H2O) gets liberated (OH group of \[NaOH~\]and hydrogen atom of NOH attached double boundedly with first carbon of nitrolic acid) and on the place of hydrogen atom (\[OH\]) sodium atom will attach and form sodium salt (\[{{O}^{-}}N{{a}^{+}}\]) such as
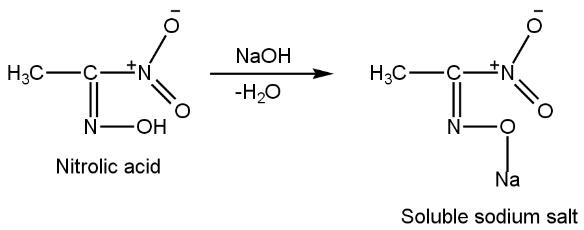
The soluble sodium salt formed is red in colour. Thus, the correct option is A.
Note: Primary nitro is one in which the \[N{{O}_{2}}\]group is attached to that carbon which is further attached to only one carbon (alkyl group) or with only one carbon chain (alkyl chain). If \[N{{O}_{2}}\]is attached to a carbon which further bonded with two carbon or with two carbon chains is known as a secondary or \[2{}^\circ \]nitro compound and if carbon is attached to three carbon further, then it is known as \[3{}^\circ \]nitro compound or tertiary nitro compound.
Complete Step by Step Solution:
Now let us take a primary nitro alkyl compound, nitro ethane (such as\[C{{H}_{3}}C{{H}_{2}}N{{O}_{2}}\]which on reacting with nitrous reagent (\[HN{{O}_{2}}\]) gives nitrolic acid (double bond between carbon and nitrogen). In this, two hydrogen of first carbon from the nitro group makes the bond with one oxygen of nitrous to release water molecule such as

Now this nitrolic acid reacts with \[NaOH~\](caustic soda) to give soluble sodium salt because after reacting nitrolic acid with \[NaOH~\], the water molecule (H2O) gets liberated (OH group of \[NaOH~\]and hydrogen atom of NOH attached double boundedly with first carbon of nitrolic acid) and on the place of hydrogen atom (\[OH\]) sodium atom will attach and form sodium salt (\[{{O}^{-}}N{{a}^{+}}\]) such as

The soluble sodium salt formed is red in colour. Thus, the correct option is A.
Note: Primary nitro is one in which the \[N{{O}_{2}}\]group is attached to that carbon which is further attached to only one carbon (alkyl group) or with only one carbon chain (alkyl chain). If \[N{{O}_{2}}\]is attached to a carbon which further bonded with two carbon or with two carbon chains is known as a secondary or \[2{}^\circ \]nitro compound and if carbon is attached to three carbon further, then it is known as \[3{}^\circ \]nitro compound or tertiary nitro compound.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




