
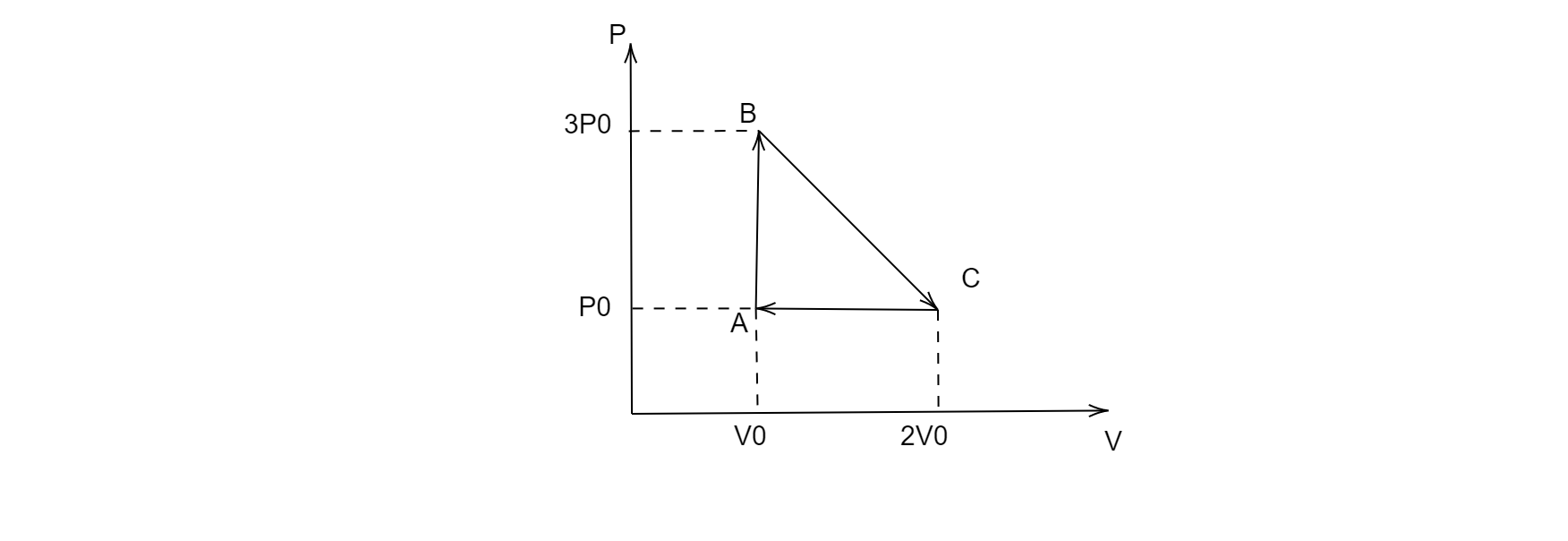
One mole of an ideal monatomic gas is taken round the cyclic process $ABCA$ as shown in the figure, calculate the heat rejected by the gas in the path $CA$ and the heat absorbed by the gas in the path $AB$.
Answer
242.1k+ views
Hint: The use of ${C_p}$ and ${C_v}$ will simplify the problem. Also, since $CA$ and $AB$ processes are isobaric and isochoric respectively, equations can be used using these two properties of the gas in the respective paths.
Complete step by step solution:
Since the gas is monatomic, therefore the value of number of moles i.e. $n$ will be $1$. The specific heat capacities at constant volume and constant pressure are:
${C_v} = \dfrac{3}{2}R$
$ \Rightarrow \dfrac{{{C_v}}}{R} = \dfrac{3}{2}$
${C_p} = \dfrac{5}{2}R$
$ \Rightarrow \dfrac{{{C_p}}}{R} = \dfrac{5}{2}$
Where ${C_p}$ and ${C_v}$ are the molar specific heat capacities of the gas at constant pressure and constant volume respectively of an ideal gas. Specific heat capacity of a substance is defined as the heat supplied per unit mass of that particular substance per unit rise in temperature.
Since the process $CA$ is isobaric i.e. the pressure remains constant throughout the process for this path, therefore the heat rejected by the gas in the path $CA$ will be:
$\Rightarrow d{h_{CA}} = {C_p}({T_{final}} - {T_{initial}})$
$\Rightarrow d{h_{CA}} = {C_p}(\dfrac{{{P_0}{V_0}}}{R} - \dfrac{{2{P_0}{V_0}}}{R})$
$\Rightarrow d{h_{CA}} = \dfrac{{{C_p}}}{R}({P_0}{V_0} - 2{P_0}{V_0})$
$\Rightarrow d{h_{CA}} = \dfrac{5}{2}({P_0}{V_0} - 2{P_0}{V_0})$
Where $d{h_{CA}}$ is the heat rejected during the process.
$\Rightarrow d{h_{CA}} = - \dfrac{5}{2}({P_0}{V_0})$
Since the process $AB$ is isochoric, i.e. the volume remains constant throughout the process for this path, therefore the heat rejected by the gas in the path $AB$ will be:
$\Rightarrow d{h_{AB}} = {C_v}dT$
Where $d{h_{AB}}$ is the heat absorbed by the monatomic gas in the process $AB$.
$\Rightarrow d{h_{AB}} = {C_v}({T_{final}} - {T_{initial}})$
$\Rightarrow {C_v}(\dfrac{{{P_f}{V_f}}}{R} - \dfrac{{{P_i}{V_i}}}{R})$
$ \Rightarrow \dfrac{{{C_v}}}{R}({P_f}{V_f} - {P_i}{V_i})$
$\Rightarrow \dfrac{3}{2}({P_f}{V_f} - {P_i}{V_i})$
$ \Rightarrow \dfrac{3}{2}(3{P_0}{V_0} - {P_0}{V_0})$
$ \Rightarrow 3{P_0}{V_0}$
Therefore, the heat rejected by the gas in the path $CA$ is $ - \dfrac{5}{2}({P_0}{V_0})$ and the heat absorbed by the gas in the path $AB$ is $3{P_0}{V_0}$.
Note: If the heat rejected in the path $CA$ is negative, then this implies that no heat is rejected in this process but there is heat absorption in the process. In the process $AB$ no such negative sign is encountered therefore the process has absorbed heat.
Complete step by step solution:
Since the gas is monatomic, therefore the value of number of moles i.e. $n$ will be $1$. The specific heat capacities at constant volume and constant pressure are:
${C_v} = \dfrac{3}{2}R$
$ \Rightarrow \dfrac{{{C_v}}}{R} = \dfrac{3}{2}$
${C_p} = \dfrac{5}{2}R$
$ \Rightarrow \dfrac{{{C_p}}}{R} = \dfrac{5}{2}$
Where ${C_p}$ and ${C_v}$ are the molar specific heat capacities of the gas at constant pressure and constant volume respectively of an ideal gas. Specific heat capacity of a substance is defined as the heat supplied per unit mass of that particular substance per unit rise in temperature.
Since the process $CA$ is isobaric i.e. the pressure remains constant throughout the process for this path, therefore the heat rejected by the gas in the path $CA$ will be:
$\Rightarrow d{h_{CA}} = {C_p}({T_{final}} - {T_{initial}})$
$\Rightarrow d{h_{CA}} = {C_p}(\dfrac{{{P_0}{V_0}}}{R} - \dfrac{{2{P_0}{V_0}}}{R})$
$\Rightarrow d{h_{CA}} = \dfrac{{{C_p}}}{R}({P_0}{V_0} - 2{P_0}{V_0})$
$\Rightarrow d{h_{CA}} = \dfrac{5}{2}({P_0}{V_0} - 2{P_0}{V_0})$
Where $d{h_{CA}}$ is the heat rejected during the process.
$\Rightarrow d{h_{CA}} = - \dfrac{5}{2}({P_0}{V_0})$
Since the process $AB$ is isochoric, i.e. the volume remains constant throughout the process for this path, therefore the heat rejected by the gas in the path $AB$ will be:
$\Rightarrow d{h_{AB}} = {C_v}dT$
Where $d{h_{AB}}$ is the heat absorbed by the monatomic gas in the process $AB$.
$\Rightarrow d{h_{AB}} = {C_v}({T_{final}} - {T_{initial}})$
$\Rightarrow {C_v}(\dfrac{{{P_f}{V_f}}}{R} - \dfrac{{{P_i}{V_i}}}{R})$
$ \Rightarrow \dfrac{{{C_v}}}{R}({P_f}{V_f} - {P_i}{V_i})$
$\Rightarrow \dfrac{3}{2}({P_f}{V_f} - {P_i}{V_i})$
$ \Rightarrow \dfrac{3}{2}(3{P_0}{V_0} - {P_0}{V_0})$
$ \Rightarrow 3{P_0}{V_0}$
Therefore, the heat rejected by the gas in the path $CA$ is $ - \dfrac{5}{2}({P_0}{V_0})$ and the heat absorbed by the gas in the path $AB$ is $3{P_0}{V_0}$.
Note: If the heat rejected in the path $CA$ is negative, then this implies that no heat is rejected in this process but there is heat absorption in the process. In the process $AB$ no such negative sign is encountered therefore the process has absorbed heat.
Recently Updated Pages
Dimensions of Charge: Dimensional Formula, Derivation, SI Units & Examples

How to Calculate Moment of Inertia: Step-by-Step Guide & Formulas

Circuit Switching vs Packet Switching: Key Differences Explained

Dimensions of Pressure in Physics: Formula, Derivation & SI Unit

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

Trending doubts
JEE Main 2026: Session 1 Results Out and Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Clemmensen and Wolff Kishner Reductions Explained for JEE & NEET

Degree of Dissociation: Meaning, Formula, Calculation & Uses

Understanding the Angle of Deviation in a Prism

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Physics Chapter 4 - Laws of Motion - 2025-26

CBSE Notes Class 11 Physics Chapter 14 - Waves - 2025-26

CBSE Notes Class 11 Physics Chapter 9 - Mechanical Properties of Fluids - 2025-26

Inductive Effect and Its Role in Acidic Strength




