Name two metals which will displace hydrogen from dilute acids:
A. ${\rm{Mg}}$and ${\rm{Zn}}$
B. ${\rm{Cu}}$and ${\rm{Au}}$
C. ${\rm{Cu}}$and ${\rm{Mg}}$
D. ${\rm{Zn}}$and ${\rm{Cu}}$
Answer
249.6k+ views
Hint: We know that a reaction where displacement of a less reactive metal from its compound occurs by a more reactive metal is termed as displacement reaction. The reactivity of metals is decided by the reactivity series of metals.
Complete step-by step answer:
Now, we discuss the condition in which displacement of hydrogen from dilute acids takes place. From dilute acids like hydrochloric acid or sulphuric acid, hydrogen is replaced by metals which are more reactive than hydrogen. The reactivity series of metals is given as follows:
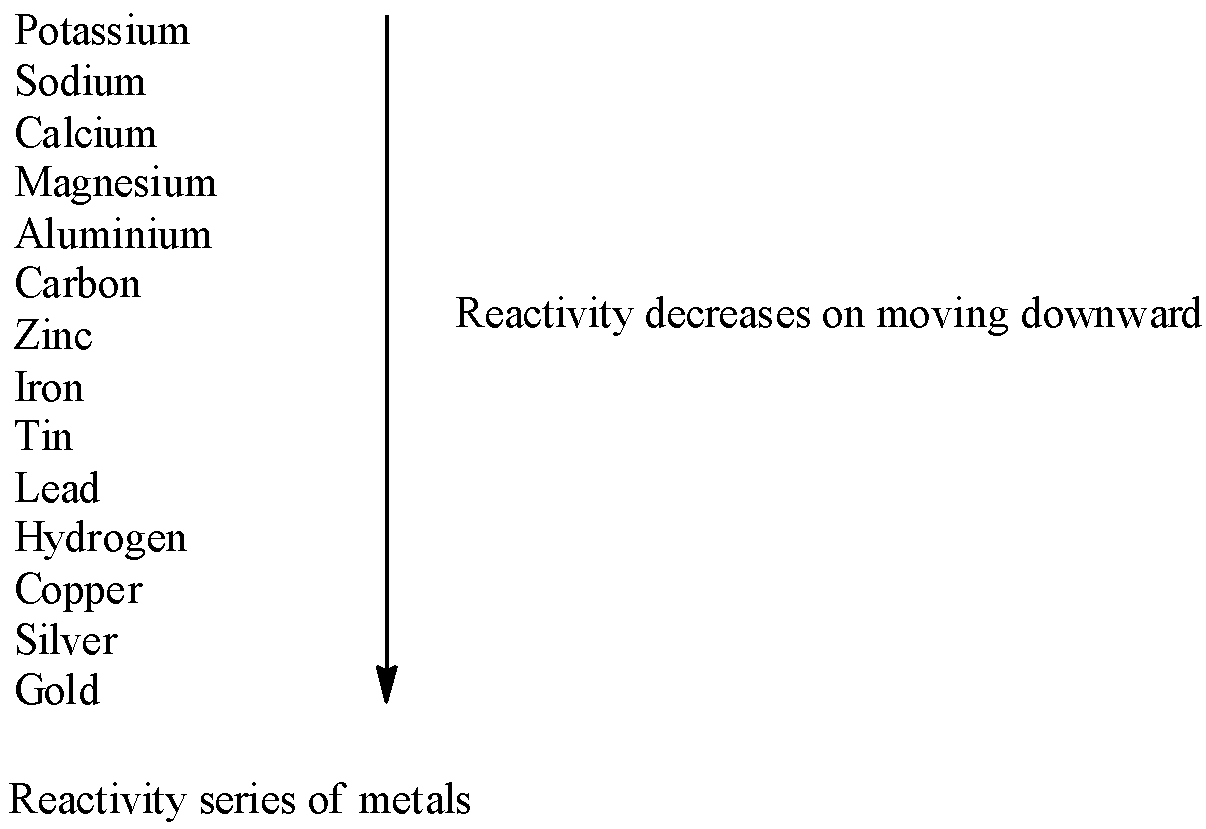
Let’s find the correct option.
Option A contains ${\rm{Mg}}$(Magnesium) and ${\rm{Zn}}$(zinc). Both magnesium and zinc are more reactive than hydrogen in the activity series. So, they displace hydrogen from dilute acids.
Option B contains ${\rm{Cu}}$(copper) and ${\rm{Au}}$(gold). Copper and gold both are less reactive than hydrogen. So, they do not displace hydrogen from dilute acids.
Option C contains ${\rm{Cu}}$(copper) and ${\rm{Mg}}$(magnesium). Copper is less reactive than hydrogen but magnesium is more reactive than hydrogen. So, displacement of hydrogen from dilute acid is possible in case of magnesium but not possible in case of copper.
Option D contains ${\rm{Zn}}$(zinc) and ${\rm{Cu}}$(copper). Copper is less reactive than hydrogen but zinc is more reactive than hydrogen. So, displacement of hydrogen from dilute acid is possible in case of zinc but not possible in case of copper.
Hence, correct option is A, that is, ${\rm{Mg}}$and ${\rm{Zn}}$
Note: Highly reactive metal like potassium, sodium and calcium displaces hydrogen from dilute acids with explosive violence, moderately reactive metals like magnesium, aluminium and zinc displaces hydrogen vigorously and metals such as copper, gold, mercury and silver do not displaces hydrogen from dilute acids.
Complete step-by step answer:
Now, we discuss the condition in which displacement of hydrogen from dilute acids takes place. From dilute acids like hydrochloric acid or sulphuric acid, hydrogen is replaced by metals which are more reactive than hydrogen. The reactivity series of metals is given as follows:

Let’s find the correct option.
Option A contains ${\rm{Mg}}$(Magnesium) and ${\rm{Zn}}$(zinc). Both magnesium and zinc are more reactive than hydrogen in the activity series. So, they displace hydrogen from dilute acids.
Option B contains ${\rm{Cu}}$(copper) and ${\rm{Au}}$(gold). Copper and gold both are less reactive than hydrogen. So, they do not displace hydrogen from dilute acids.
Option C contains ${\rm{Cu}}$(copper) and ${\rm{Mg}}$(magnesium). Copper is less reactive than hydrogen but magnesium is more reactive than hydrogen. So, displacement of hydrogen from dilute acid is possible in case of magnesium but not possible in case of copper.
Option D contains ${\rm{Zn}}$(zinc) and ${\rm{Cu}}$(copper). Copper is less reactive than hydrogen but zinc is more reactive than hydrogen. So, displacement of hydrogen from dilute acid is possible in case of zinc but not possible in case of copper.
Hence, correct option is A, that is, ${\rm{Mg}}$and ${\rm{Zn}}$
Note: Highly reactive metal like potassium, sodium and calcium displaces hydrogen from dilute acids with explosive violence, moderately reactive metals like magnesium, aluminium and zinc displaces hydrogen vigorously and metals such as copper, gold, mercury and silver do not displaces hydrogen from dilute acids.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




