In which of the following species the interatomic bond angle is \[109^\circ 28\prime \;\].
A.\[N{H_3},{\rm{ }}{\left( {B{F_4}} \right)^{ - 1}}\;\]
B.\[{\left( {N{H_4}} \right)^ + },{\rm{ }}B{F_3}\]
C.\[N{H_3},{\rm{ }}B{F_4}\]
D.\[{\left( {N{H_2}} \right)^{ - 1}},{\rm{ }}B{F_3}\]
Answer
248.7k+ views
Hint: Bond angle 109°28′
is demonstrated by a regular tetrahedral molecular geometry.
When the central metal atom having a tetrahedral shape has 4 bond pairs and no lone pairs, it will demonstrate this bond angle.
Complete step by step solution:The angle between two bonds arising from the same atom in a covalent species is comprehended as the bond angle.
The given bond angle is in species with regular tetrahedral geometry and they carry a hybridization of\[s{p^3}\].
For this, the steric number is 4. The steric number is the no.of atoms, groups, or lone pairs for the central metal atom.
For the given bond angle, there are generally 4 bonding pairs of electrons and no lone pairs.
So currently we have to study the provided molecules and attempt to find out the number of bond pairs in them.
Out of the given options, \[N{H_3},{\rm{ }}{\left( {B{F_4}} \right)^{ - 1}}\;\]both have interatomic bond angles of \[109^\circ 28\prime \;\].
\[N{H_3}\;\]
There is an N atom which is the central atom. It has 5 valence electrons.
Three electrons undergo bond formation with each of the three hydrogen atoms and the rest lone pair of electrons is there.
So, there are three bond pairs and one lone pair.
So, it has a pyramidal structure due to the repulsion between bond pairs and the lone pair.
So, the bond angle is \[109^\circ 28\prime \;\].
\[{\left( {B{F_4}} \right)^{ - 1}}\;\]
The shape is tetrahedral and, in the molecule, there are four bond pairs and zero lone pairs.
There are four B-F bonds.
So, the bond angle is \[109^\circ 28\prime \;\].
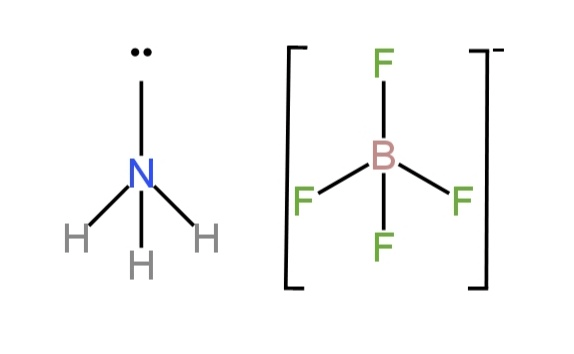
Image: Structure of \[N{H_3},{\rm{ }}{\left( {B{F_4}} \right)^{ - 1}}\;\]
So, option A is correct.
Note: Ammonia is one of the central components in a ton of household cleaning products.
It is utilized as a cleaning agent and can be utilized to peel off stains from mirrors, tubs, sinks, windows, and more. Some other benefits are antimicrobial agents or antiseptics, and ammonia is also utilized as a fuel.
is demonstrated by a regular tetrahedral molecular geometry.
When the central metal atom having a tetrahedral shape has 4 bond pairs and no lone pairs, it will demonstrate this bond angle.
Complete step by step solution:The angle between two bonds arising from the same atom in a covalent species is comprehended as the bond angle.
The given bond angle is in species with regular tetrahedral geometry and they carry a hybridization of\[s{p^3}\].
For this, the steric number is 4. The steric number is the no.of atoms, groups, or lone pairs for the central metal atom.
For the given bond angle, there are generally 4 bonding pairs of electrons and no lone pairs.
So currently we have to study the provided molecules and attempt to find out the number of bond pairs in them.
Out of the given options, \[N{H_3},{\rm{ }}{\left( {B{F_4}} \right)^{ - 1}}\;\]both have interatomic bond angles of \[109^\circ 28\prime \;\].
\[N{H_3}\;\]
There is an N atom which is the central atom. It has 5 valence electrons.
Three electrons undergo bond formation with each of the three hydrogen atoms and the rest lone pair of electrons is there.
So, there are three bond pairs and one lone pair.
So, it has a pyramidal structure due to the repulsion between bond pairs and the lone pair.
So, the bond angle is \[109^\circ 28\prime \;\].
\[{\left( {B{F_4}} \right)^{ - 1}}\;\]
The shape is tetrahedral and, in the molecule, there are four bond pairs and zero lone pairs.
There are four B-F bonds.
So, the bond angle is \[109^\circ 28\prime \;\].

Image: Structure of \[N{H_3},{\rm{ }}{\left( {B{F_4}} \right)^{ - 1}}\;\]
So, option A is correct.
Note: Ammonia is one of the central components in a ton of household cleaning products.
It is utilized as a cleaning agent and can be utilized to peel off stains from mirrors, tubs, sinks, windows, and more. Some other benefits are antimicrobial agents or antiseptics, and ammonia is also utilized as a fuel.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Derivation of Equation of Trajectory Explained for Students

Understanding the Angle of Deviation in a Prism

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




