In the following reaction sequence:
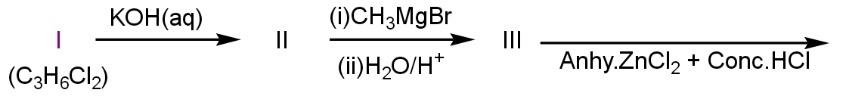
give turbidity immediately
(A)
(B)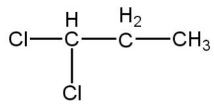
(C)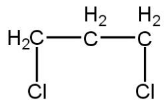
(D)
Answer
255.3k+ views
Hint: Turbidity in terms of the Lucas test is the measure to differentiate primary, secondary and tertiary alcohol when reacts with the Lucas reagent \[\left( Anhy.ZnC{{l}_{2}}+\text{ }Conc.HCl \right)\]. Turbidity (cloudiness) will immediately appear in the case of three-degree alcohol (tertiary). We need to reduce the turbidity of drinking water to get healthy purified water.
Complete Step by Step Solution:
Compound A on hydrolysis by \[KOH\text{ }\left( aq \right)\] gives compound \[propane-1,1-diol\]. This compound further releases a water molecule and after the attack of the Grignard reagent gives primary alcohol.
Compound B also gives primary alcohol after hydrolysis \[\left( give\text{ }propane-1,3-diol \right)\]and further reacts with the Grignard reagent.
Compound C which when reacting with aqueous KOH gets hydrolyzed and gives \[propane-2,2-diol\] (unstable) in step 1. This compound further releases a water molecule to give a ketone which is in equilibrium with the compound formed in the first step. This ketone, further attacked by Grignard reagent (negative part,\[C{{H}_{3}}\] of Grignard reagent attack ketonic carbon, positively charged) and oxygen (negatively charged)of ketone group get protonated in presence of water molecule to give the hydroxyl group (\[OH\]group) such as
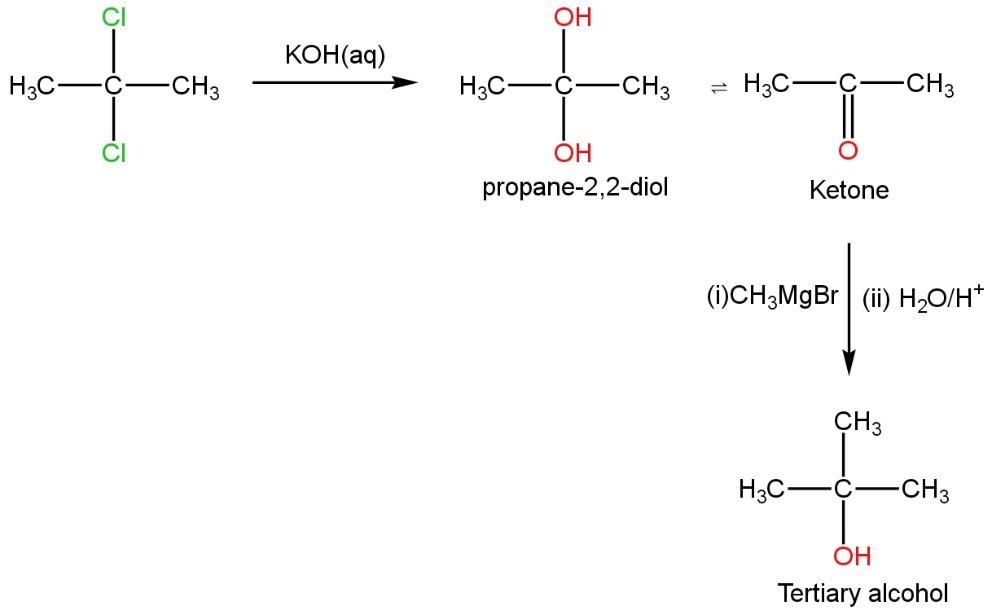
This results in the formation of three-degree alcohol (tertiary alcohol). This three-degree alcohol immediately gives turbidity under the action of Lucas’s reagent.
Thus, the correct option is C.
Note: Aqueous\[KOH\]is used to hydrolysis another compound as\[KOH\]is a strong nucleophile reagent. In any halogen-containing compound, halogens are good leaving groups and thus leave the compound due to its large size and high electronegativity value. As at least the compound, carbonation generated and thus attacked by\[O{{H}^{-}}\] (nucleophile), hydroxyl ion and hydrolysis take place. Grignard reagent molecular formula is\[RMgX\], where R is an alkyl group and X is a halogen. In any aqueous solution Grignard reagent breaks into negative (R) and positive (MgX) ions.
Complete Step by Step Solution:
Compound A on hydrolysis by \[KOH\text{ }\left( aq \right)\] gives compound \[propane-1,1-diol\]. This compound further releases a water molecule and after the attack of the Grignard reagent gives primary alcohol.
Compound B also gives primary alcohol after hydrolysis \[\left( give\text{ }propane-1,3-diol \right)\]and further reacts with the Grignard reagent.
Compound C which when reacting with aqueous KOH gets hydrolyzed and gives \[propane-2,2-diol\] (unstable) in step 1. This compound further releases a water molecule to give a ketone which is in equilibrium with the compound formed in the first step. This ketone, further attacked by Grignard reagent (negative part,\[C{{H}_{3}}\] of Grignard reagent attack ketonic carbon, positively charged) and oxygen (negatively charged)of ketone group get protonated in presence of water molecule to give the hydroxyl group (\[OH\]group) such as

This results in the formation of three-degree alcohol (tertiary alcohol). This three-degree alcohol immediately gives turbidity under the action of Lucas’s reagent.
Thus, the correct option is C.
Note: Aqueous\[KOH\]is used to hydrolysis another compound as\[KOH\]is a strong nucleophile reagent. In any halogen-containing compound, halogens are good leaving groups and thus leave the compound due to its large size and high electronegativity value. As at least the compound, carbonation generated and thus attacked by\[O{{H}^{-}}\] (nucleophile), hydroxyl ion and hydrolysis take place. Grignard reagent molecular formula is\[RMgX\], where R is an alkyl group and X is a halogen. In any aqueous solution Grignard reagent breaks into negative (R) and positive (MgX) ions.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




