In potassium, the order of energy levels is:
(A) $3s > 3d$
(B) $4s < 3d$
(C) $4s > 4p$
(D) $4s = 3d$
Answer
255.6k+ views
Hint: The electronic configuration of potassium is $[Ar]4{s^1}$. Aufbau principle states that in the ground state of atoms, the orbitals are filled in the order of their increasing energies.
Complete step by step solution:
According to the Aufbau principle, the electrons first occupy the lowest energy orbital available to them and enter into higher energy orbitals only when the lower energy orbitals are filled. The order in which the energies of the orbitals increase and also the order in which the orbitals are filled are $1s,2s,2p,3s,3p,4s,3d,5s,4d,5p,6s,......$
This can be easily understood by the following diagram:
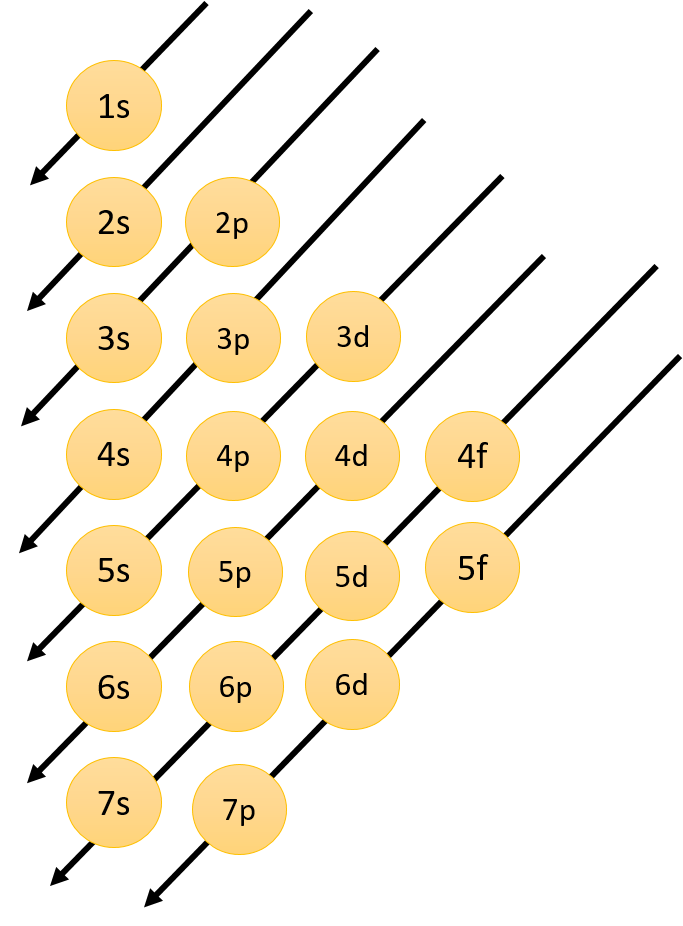
The energy of the orbitals depends upon the principal quantum number ($n$) and azimuthal quantum number ($l$). Orbital energy increases as ($n + l$) increases. So lower the value of ($n + l$) for an orbital, the lower is its energy. If there are two orbitals with the same value of ($n + l$) the one with the smaller value of $n$ has the lower energy.
Therefore a subshell with a lower value of ($n + l$) possess lower energy and is filled first. Let us consider $4s$ and $3d$ subshell.
For $4s$ subshell, $n + l = 4 + 0 = 4$,
while for 3d subshell, $n + l = 3 + 2 = 5$.
Hence, the ($n + l$) value of $4s$ subshell is lower than that of $3d$ subshell, so its energy is lower than that of $3d$ subshell. Thus, the $4s$ subshell is filled before the $3d$ subshell.
In the case of potassium ($K$) (atomic number ($Z$) =$19$), the electronic configuration is $1{s^2}2{s^2}2{p^6}3{s^2}3{p^6}4{s^1}$. From the electronic configuration, it can be observed that the last electron of the potassium entered into the $4s$ subshell instead of the $3d$ subshell. This is because the energy of $4s$ subshell is lower than that of $3d$ subshell as illustrated above.
Hence, option (B) is the correct answer.
Note: The energies of different orbitals drop to a different extent with an increase in atomic number because of the effective nuclear charge. The half-filled and completely filled subshells possess extra stability due to the symmetrical distribution of electrons and greater exchange energy. That is why the electronic configuration of some elements are exceptional and not in accordance with the normal order of filling. E.g., Cr ($Z = 24$) and Cu ($Z = 29$).
Complete step by step solution:
According to the Aufbau principle, the electrons first occupy the lowest energy orbital available to them and enter into higher energy orbitals only when the lower energy orbitals are filled. The order in which the energies of the orbitals increase and also the order in which the orbitals are filled are $1s,2s,2p,3s,3p,4s,3d,5s,4d,5p,6s,......$
This can be easily understood by the following diagram:

The energy of the orbitals depends upon the principal quantum number ($n$) and azimuthal quantum number ($l$). Orbital energy increases as ($n + l$) increases. So lower the value of ($n + l$) for an orbital, the lower is its energy. If there are two orbitals with the same value of ($n + l$) the one with the smaller value of $n$ has the lower energy.
Therefore a subshell with a lower value of ($n + l$) possess lower energy and is filled first. Let us consider $4s$ and $3d$ subshell.
For $4s$ subshell, $n + l = 4 + 0 = 4$,
while for 3d subshell, $n + l = 3 + 2 = 5$.
Hence, the ($n + l$) value of $4s$ subshell is lower than that of $3d$ subshell, so its energy is lower than that of $3d$ subshell. Thus, the $4s$ subshell is filled before the $3d$ subshell.
In the case of potassium ($K$) (atomic number ($Z$) =$19$), the electronic configuration is $1{s^2}2{s^2}2{p^6}3{s^2}3{p^6}4{s^1}$. From the electronic configuration, it can be observed that the last electron of the potassium entered into the $4s$ subshell instead of the $3d$ subshell. This is because the energy of $4s$ subshell is lower than that of $3d$ subshell as illustrated above.
Hence, option (B) is the correct answer.
Note: The energies of different orbitals drop to a different extent with an increase in atomic number because of the effective nuclear charge. The half-filled and completely filled subshells possess extra stability due to the symmetrical distribution of electrons and greater exchange energy. That is why the electronic configuration of some elements are exceptional and not in accordance with the normal order of filling. E.g., Cr ($Z = 24$) and Cu ($Z = 29$).
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




