In Carius’ method of estimation of halogen, 0.172 g of an organic compound showed the presence of 0.08 g of bromine. Which of these is the correct structure of the compound?
A.
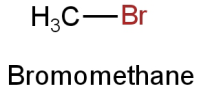
B.
C.
D.
Answer
256.2k+ views
Hint: Carius method is a procedure used to estimate the percentage of halogen in an organic compound.
In this method, a halogen-containing organic compound is heated in the presence of silver nitrate.
Formula Used:
Percentage of halogen =
\[\dfrac{{{\rm{the atomic mass of bromine}}}}{{{\rm{mass of the organic compound}}}}{\rm{ }}\].×100
Complete Step by Step Solution:
The mass of the organic compound = 0.172g
Mass of bromine present in the organic compound = 0.08g
So, the percentage of bromine =\[\dfrac{{0.08}}{{0.172}}(100)\] \[ = 46.51\% \].
The atomic mass of bromine = 80g.
A.
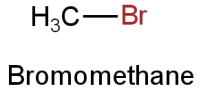
Image: Option A
This is bromomethane.
Molar mass
\[{\rm{ = [12 + 3(1) + 80]g}}\]
\[{\rm{ = 95g}}\]
The molar mass of bromoethane is 95g.
There is one bromine atom.
So, it contains 80g of bromine.
So, the percentage of bromine
\[ = \dfrac{{80}}{{95}} \times 100 = 84.21\% \]
This doesn't resemble the percentage of bromine in the given organic compound.
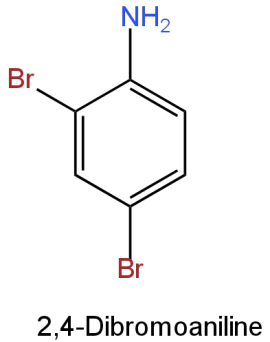
B.
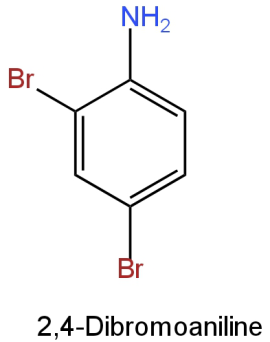
Image: Option B
This is 2,4-dibromoaniline (\[{{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{5}}}{\rm{B}}{{\rm{r}}_{\rm{2}}}{\rm{N}}\])
Molar mass \[{\rm{ = [12(6) + 5(1) + 2(80) + 14]g}}\] \[{\rm{ = 251g}}\]
It contains 2 bromine atoms.
So, the percentage of bromine
\[\begin{array}{*{20}{l}}{ = {\rm{ }}\left[ {\dfrac{{2\left( {80} \right)}}{{251}}} \right] \times 100}\\{ = {\rm{ }}63.74\% }\end{array}\]
This doesn't resemble the percentage of bromine in the given organic compound.
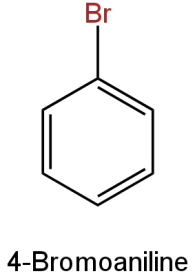
C.
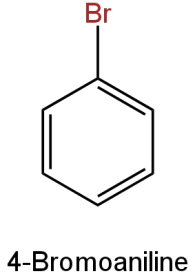
Image: Option C
This is 4-bromoaniline(\[{{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{6}}}{\rm{BrN}}\]).
Molar mass
\[{\rm{ = }}\left[ {{\rm{12}}\left( {\rm{6}} \right){\rm{ + 6}}\left( {\rm{1}} \right){\rm{ + }}\left( {{\rm{80}}} \right){\rm{ + 14}}} \right]{\rm{g}}\]
\[{\rm{ = 172g}}\]
It contains one bromine atom.
Percentage of bromine
\[ = \left( {\dfrac{{80}}{{172}}} \right) \times 100\]
\[ = 46.51\% \]
This is the same percentage as the bromine in the given organic compound.
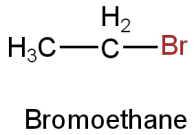
D.
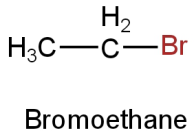
Image: Option D
This is bromoethane (\[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{Br}}\]).
Molar mass \[{\rm{ = }}\left[ {{\rm{2}}\left( {{\rm{12}}} \right){\rm{ + 5}}\left( {\rm{1}} \right){\rm{ + 80}}} \right]{\rm{g}}\] \[{\rm{ = 109g}}\]
Only one bromine atom is present.
So, the percentage of bromine
\[ = {\rm{ }}\left[ {\left( {\dfrac{{80}}{{109}}} \right) \times 100} \right]\] \[ = 73.39\% \]
This doesn't resemble the percentage of bromine in the given organic compound.
So, option C is correct.
Additional Information:In the Carius method, a known mass of an organic compound is heated with fuming nitric acid in the presence of silver nitrate contained in a hard glass tube known as the Carius tube. Carbon and hydrogen that are present in the compound are oxidised to carbon dioxide and water.
The halogen present forms the corresponding silver halide.
It is filtered, washed, dried and weighed.
Note: While attending to the question, one must calculate the percentage of bromine present in each given option. He/she should have calculated the percentage of bromine in the given organic compound first. Then, after matching the bromine percentage in the given organic compound with the bromine percentage of individual options, the correct organic compound will be determined.
In this method, a halogen-containing organic compound is heated in the presence of silver nitrate.
Formula Used:
Percentage of halogen =
\[\dfrac{{{\rm{the atomic mass of bromine}}}}{{{\rm{mass of the organic compound}}}}{\rm{ }}\].×100
Complete Step by Step Solution:
The mass of the organic compound = 0.172g
Mass of bromine present in the organic compound = 0.08g
So, the percentage of bromine =\[\dfrac{{0.08}}{{0.172}}(100)\] \[ = 46.51\% \].
The atomic mass of bromine = 80g.
A.
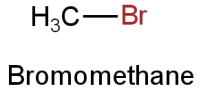
Image: Option A
This is bromomethane.
Molar mass
\[{\rm{ = [12 + 3(1) + 80]g}}\]
\[{\rm{ = 95g}}\]
The molar mass of bromoethane is 95g.
There is one bromine atom.
So, it contains 80g of bromine.
So, the percentage of bromine
\[ = \dfrac{{80}}{{95}} \times 100 = 84.21\% \]
This doesn't resemble the percentage of bromine in the given organic compound.
B.

Image: Option B
This is 2,4-dibromoaniline (\[{{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{5}}}{\rm{B}}{{\rm{r}}_{\rm{2}}}{\rm{N}}\])
Molar mass \[{\rm{ = [12(6) + 5(1) + 2(80) + 14]g}}\] \[{\rm{ = 251g}}\]
It contains 2 bromine atoms.
So, the percentage of bromine
\[\begin{array}{*{20}{l}}{ = {\rm{ }}\left[ {\dfrac{{2\left( {80} \right)}}{{251}}} \right] \times 100}\\{ = {\rm{ }}63.74\% }\end{array}\]
This doesn't resemble the percentage of bromine in the given organic compound.
C.

Image: Option C
This is 4-bromoaniline(\[{{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{6}}}{\rm{BrN}}\]).
Molar mass
\[{\rm{ = }}\left[ {{\rm{12}}\left( {\rm{6}} \right){\rm{ + 6}}\left( {\rm{1}} \right){\rm{ + }}\left( {{\rm{80}}} \right){\rm{ + 14}}} \right]{\rm{g}}\]
\[{\rm{ = 172g}}\]
It contains one bromine atom.
Percentage of bromine
\[ = \left( {\dfrac{{80}}{{172}}} \right) \times 100\]
\[ = 46.51\% \]
This is the same percentage as the bromine in the given organic compound.
D.

Image: Option D
This is bromoethane (\[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{Br}}\]).
Molar mass \[{\rm{ = }}\left[ {{\rm{2}}\left( {{\rm{12}}} \right){\rm{ + 5}}\left( {\rm{1}} \right){\rm{ + 80}}} \right]{\rm{g}}\] \[{\rm{ = 109g}}\]
Only one bromine atom is present.
So, the percentage of bromine
\[ = {\rm{ }}\left[ {\left( {\dfrac{{80}}{{109}}} \right) \times 100} \right]\] \[ = 73.39\% \]
This doesn't resemble the percentage of bromine in the given organic compound.
So, option C is correct.
Additional Information:In the Carius method, a known mass of an organic compound is heated with fuming nitric acid in the presence of silver nitrate contained in a hard glass tube known as the Carius tube. Carbon and hydrogen that are present in the compound are oxidised to carbon dioxide and water.
The halogen present forms the corresponding silver halide.
It is filtered, washed, dried and weighed.
Note: While attending to the question, one must calculate the percentage of bromine present in each given option. He/she should have calculated the percentage of bromine in the given organic compound first. Then, after matching the bromine percentage in the given organic compound with the bromine percentage of individual options, the correct organic compound will be determined.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




