In acidic and alkaline solution amino acid exists as a-
(A) Positive and negative ion respectively
(B) Negative and positive ion respectively
(C) Neutral in both medium
(D) None of these.
Answer
251.7k+ views
Hint: In acidic conditions, the amino acid acts as a base and accepts a proton at the amino group. In alkaline conditions, the amino acid acts as an acid and donates a proton from its carboxyl group.
Complete step by step solution:
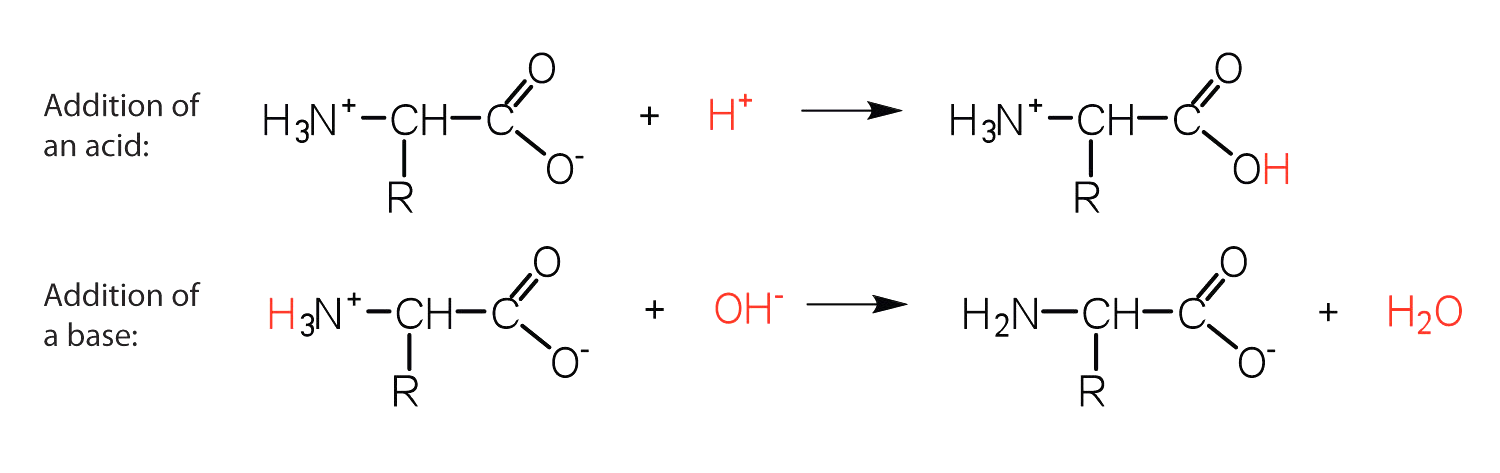
-The structure of an amino acid allows it to act as both an acid and a base. This is because at a certain pH value (different for each amino acid) nearly all the amino acid molecules exist as zwitterions.
-If acid is added to a solution containing the zwitterion, the carboxylate group captures a hydrogen $\left(\mathrm{H}^{+}\right)$ ion and the amino acid becomes positively charged.
-If base is added, ion removal of the $\mathrm{H}^{+}$ ion from the amino group of the zwitterion produces a negatively charged amino acid.
In both circumstances, the amino acid acts to maintain the $\mathrm{pH}$ of the system- that is, to remove the added acid ( $\mathrm{H}^{+}$ ) or base ( $\mathrm{OH}^{-}$ ) from solution.
-Thus, in acidic medium, the amino acid has a positive charge, thus a positive charge. In a basic medium, it possesses a negative charge.
Clearly, the answer is A.
Note: Zwitterion is a molecule that contains an equal number of positively- and negatively-charged functional groups. In an acidic and basic medium, it possesses a charge and becomes ions.
Complete step by step solution:
-The structure of an amino acid allows it to act as both an acid and a base. This is because at a certain pH value (different for each amino acid) nearly all the amino acid molecules exist as zwitterions.
-If acid is added to a solution containing the zwitterion, the carboxylate group captures a hydrogen $\left(\mathrm{H}^{+}\right)$ ion and the amino acid becomes positively charged.
-If base is added, ion removal of the $\mathrm{H}^{+}$ ion from the amino group of the zwitterion produces a negatively charged amino acid.
In both circumstances, the amino acid acts to maintain the $\mathrm{pH}$ of the system- that is, to remove the added acid ( $\mathrm{H}^{+}$ ) or base ( $\mathrm{OH}^{-}$ ) from solution.
-Thus, in acidic medium, the amino acid has a positive charge, thus a positive charge. In a basic medium, it possesses a negative charge.

Clearly, the answer is A.
Note: Zwitterion is a molecule that contains an equal number of positively- and negatively-charged functional groups. In an acidic and basic medium, it possesses a charge and becomes ions.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




