Formula for the following compound Tin (IV) oxide is $SnO_2$:
(A) True
(B) False
Answer
265.8k+ views
Hint: We should know that Tin dioxide is a tin oxide compound which consists of Tin (IV) covalently bound to two oxygen atoms. The chemical formula of Tin (IV) oxide, will further be understood from the structure of Tin (IV) oxide.
Complete step by step answer:
> Tin (IV) oxide is also known as Stannic Oxide. We know that the symbol of Tin is Sn.
Here is the structure of Tin (IV) oxide, for better understanding of the formula:
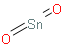
> The Tin (IV) crystallizes with the rutile structure. As such the Tin atoms are 6 coordinate and the oxygen atoms are 3 coordinate. ${\text{Sn}}{{\text{O}}_{\text{2}}}$ is usually regarded as an oxygen deficient n type semiconductor. Hydrous forms of ${\text{Sn}}{{\text{O}}_{\text{2}}}$ has been described as stannic acid.
> The bond formation of Tin and the two oxygen atoms are known as just polar covalent. This is because of the electronegativity difference between Tin (1.8) and that of Oxygen (3.5). So, with a difference of only 1.7 in electronegativity, ionic bonds cannot be formed.
> The chemical formula of Tin (IV) oxide is ${\text{Sn}}{{\text{O}}_{\text{2}}}$.
Therefore, the answer to the question that the formula for the following compound Tin (IV) oxide is True.
So Option A is the correct answer to the mentioned question.
- Tin (IV) oxide appears as white or off white crystalline solid or powder. It is insoluble in water and soluble in concentrated sulphuric acid and hydrochloric acid. It occurs in nature as a mineral cassiterite. It is used as a catalyst in putty as a polishing powder for steel and glass, in ceramic places and colours.
Note: As mentioned earlier ${\text{Sn}}{{\text{O}}_{\text{2}}}$ is insoluble in water. But it is also amphoteric in nature. By amphoteric we mean, it dissolves in acids and bases. It is also diamagnetic in nature.
Complete step by step answer:
> Tin (IV) oxide is also known as Stannic Oxide. We know that the symbol of Tin is Sn.
Here is the structure of Tin (IV) oxide, for better understanding of the formula:
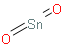
> The Tin (IV) crystallizes with the rutile structure. As such the Tin atoms are 6 coordinate and the oxygen atoms are 3 coordinate. ${\text{Sn}}{{\text{O}}_{\text{2}}}$ is usually regarded as an oxygen deficient n type semiconductor. Hydrous forms of ${\text{Sn}}{{\text{O}}_{\text{2}}}$ has been described as stannic acid.
> The bond formation of Tin and the two oxygen atoms are known as just polar covalent. This is because of the electronegativity difference between Tin (1.8) and that of Oxygen (3.5). So, with a difference of only 1.7 in electronegativity, ionic bonds cannot be formed.
> The chemical formula of Tin (IV) oxide is ${\text{Sn}}{{\text{O}}_{\text{2}}}$.
Therefore, the answer to the question that the formula for the following compound Tin (IV) oxide is True.
So Option A is the correct answer to the mentioned question.
- Tin (IV) oxide appears as white or off white crystalline solid or powder. It is insoluble in water and soluble in concentrated sulphuric acid and hydrochloric acid. It occurs in nature as a mineral cassiterite. It is used as a catalyst in putty as a polishing powder for steel and glass, in ceramic places and colours.
Note: As mentioned earlier ${\text{Sn}}{{\text{O}}_{\text{2}}}$ is insoluble in water. But it is also amphoteric in nature. By amphoteric we mean, it dissolves in acids and bases. It is also diamagnetic in nature.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




