Find the number of $P-O$ bonds of identical length in hypophosphate ion.
Answer
267.3k+ views
Hint: Draw the structure of hypophosphate ion and then count the number of $P-O$ bonds in it which are of identical length.
Complete Step by Step Answer:
The oxyanion of Hypophosphoric acid ${{H}_{2}}P{{O}_{3}}$ ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is hypophosphate ion(${{P}_{2}}{{O}_{6}}^{4-}$). The structure of this ion has been a subject of speculation and controversy since hypophosphoric acid was discovered by Salzer in 1877 . Bell and Sugden showed conclusively that the salts which they studied were not derived from an acid of simple structure like ${{H}_{2}}P{{O}_{3}}$ and of the possible dimeric formulae for the acid . In this way the structure for this hypophosphate ion is represented finally as:
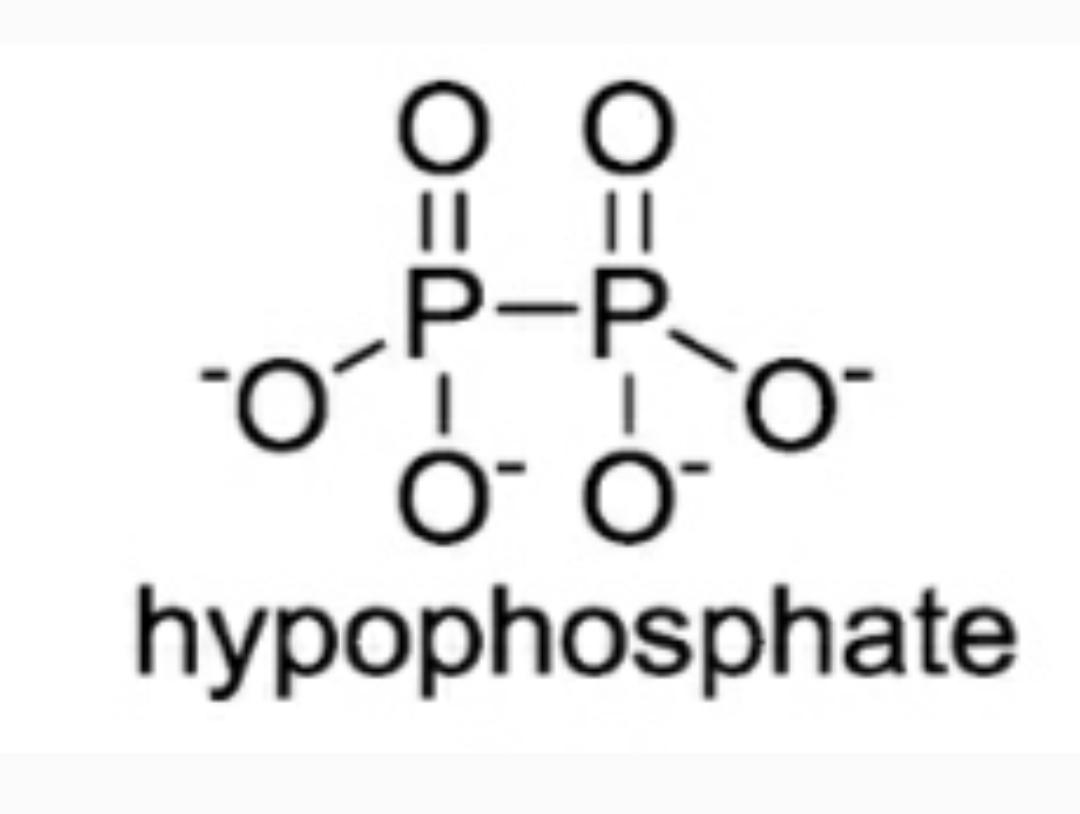
Now since here in this structure there are negative charges present and double bonds on oxygen atoms, all atomic bonds between P and O( $P-O$) do resonate. As we know that in resonance all the bond lengths are of identical length due to the regular shift of double bond in each bond respectively. Thus , if we count the number of these bonds then we get the total number as 6.
Thus , the correct answer is 6.
Note: It should be known that when negative charge is present on an electronegative ion like that on oxygen and double bonds are present then resonance occurs and due to resonance only the total number of bonds will be 6. Some students take into account only the single bonds of $P-O$ and declare the final answer as 4 which is the wrong answer.
Complete Step by Step Answer:
The oxyanion of Hypophosphoric acid ${{H}_{2}}P{{O}_{3}}$ ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is hypophosphate ion(${{P}_{2}}{{O}_{6}}^{4-}$). The structure of this ion has been a subject of speculation and controversy since hypophosphoric acid was discovered by Salzer in 1877 . Bell and Sugden showed conclusively that the salts which they studied were not derived from an acid of simple structure like ${{H}_{2}}P{{O}_{3}}$ and of the possible dimeric formulae for the acid . In this way the structure for this hypophosphate ion is represented finally as:

Now since here in this structure there are negative charges present and double bonds on oxygen atoms, all atomic bonds between P and O( $P-O$) do resonate. As we know that in resonance all the bond lengths are of identical length due to the regular shift of double bond in each bond respectively. Thus , if we count the number of these bonds then we get the total number as 6.
Thus , the correct answer is 6.
Note: It should be known that when negative charge is present on an electronegative ion like that on oxygen and double bonds are present then resonance occurs and due to resonance only the total number of bonds will be 6. Some students take into account only the single bonds of $P-O$ and declare the final answer as 4 which is the wrong answer.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




