When ethylene bromide is treated with \[Zn\] we get
(a) Alkane
(b) Alkene
(c) Alkyne
(d) All
Answer
259.2k+ views
Hint: When two atoms or substituents which are connected to two neighboring carbon atoms undergo elimination via a one or two-step reaction pathway. Such reactions are recognized as elimination reactions.
Complete Step by Step Answer:
The given question represents the reaction of \[Zn\]with ethylene bromide.
The structure of ethylene bromide contains two bromine atoms which are seated on two adjacent carbon atoms. The structure of ethylene bromide can be represented below.
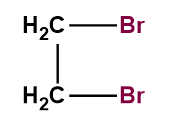
Figure: Structure of ethylene bromide.
When the alcoholic solution of ethylene bromide is treated with \[Zn\], the formation of ethylene molecules is observed with the formation of \[ZnB{r_2}\]salt.
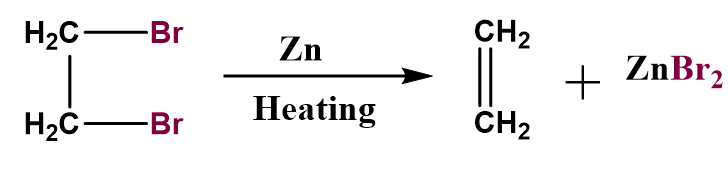
Figure: Reaction of ethylene bromide with zinc.
In the above reaction the \[Zn\] behave as a de-halogenating or eliminating agent i.e., it causes the removal of adjacent bromine atoms on neighboring carbon atoms.
The question contains options such as an alkane, alkene, and alkyne. Hence, from the above explanation, it is clear that when ethylene bromide is heated with zinc the formation of ethylene (alkene) occurs. There is no alkane and alkyne formation observed.
Therefore, the option which contained alkane (a), alkyne (c) and mixture (d) is wrong.
Therefore, from the above discussion, it is quite clear that, option (b) will be the correct answer.
Note: The alkane, alkene and alkyne are saturated (alkane) and unsaturated (alkene and alkyne) hydrocarbons. Alkane is a saturated hydrocarbon, in which the carbon atoms contain the single bonds Alkene and alkyne belong to the family of unsaturated hydrocarbons in which the double and triple bond is present between the carbon atoms.
Complete Step by Step Answer:
The given question represents the reaction of \[Zn\]with ethylene bromide.
The structure of ethylene bromide contains two bromine atoms which are seated on two adjacent carbon atoms. The structure of ethylene bromide can be represented below.

Figure: Structure of ethylene bromide.
When the alcoholic solution of ethylene bromide is treated with \[Zn\], the formation of ethylene molecules is observed with the formation of \[ZnB{r_2}\]salt.

Figure: Reaction of ethylene bromide with zinc.
In the above reaction the \[Zn\] behave as a de-halogenating or eliminating agent i.e., it causes the removal of adjacent bromine atoms on neighboring carbon atoms.
The question contains options such as an alkane, alkene, and alkyne. Hence, from the above explanation, it is clear that when ethylene bromide is heated with zinc the formation of ethylene (alkene) occurs. There is no alkane and alkyne formation observed.
Therefore, the option which contained alkane (a), alkyne (c) and mixture (d) is wrong.
Therefore, from the above discussion, it is quite clear that, option (b) will be the correct answer.
Note: The alkane, alkene and alkyne are saturated (alkane) and unsaturated (alkene and alkyne) hydrocarbons. Alkane is a saturated hydrocarbon, in which the carbon atoms contain the single bonds Alkene and alkyne belong to the family of unsaturated hydrocarbons in which the double and triple bond is present between the carbon atoms.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




