$(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ gives:
A. $3 - $hexyne
B. $2 - $hexyne
C. $2,3 - $ hexadiene
D. $2,4 - $hexadiene
Answer
260.1k+ views
Hint : We can get the product of the given reaction with the help of Saytzeff’s rule which is used in analysis of elimination reactions of organic chemistry. Elimination reaction of any halides and alcohol produces different alkenes. In such a type of reaction Saytzeff’s rule is used to predict major products. According to this rule, stable alkene is formed if the removal of hydrogen from $\beta - $ carbon which has a low number of substituents. This rule is applicable for dehydrohalogenation reactions. This problem is also based on dehydrobromination of the reactant so the product will be formed with the help of Saytzeff's rule.
Complete step by step solution:
So we can say that according to the Satzev’s rule $(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ it produce $3 - $hexyne. It undergoes dehydrobromination where a molecule of hydrobromide gets eliminated from $(E) - 3 - $bromo$ - 3 - $hexene. A dehydrobromination reaction is a kind of elimination reaction where elimination of hydrogen bromide from a substrate takes place. This reaction is mainly associated with the synthesis of alkenes. The reaction of $(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ is given below.
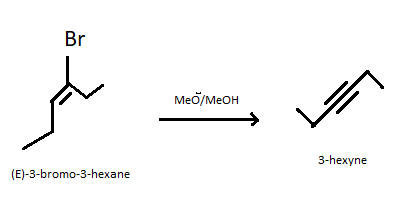
Hence option A is correct that is $(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ gives $3 - $hexyne.
Note : We have approached this reaction with the help of saytzeff rule which favours the alkene with less number of hydrogen on double bonded carbon atoms.As the reaction is dehydrobromination so saytzeff rule is applicable here. We know that during the elimination reaction proton is removed from that carbon atom which have less number of substituents.Therefore $(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ gives $3 - $hexyne.
Complete step by step solution:
So we can say that according to the Satzev’s rule $(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ it produce $3 - $hexyne. It undergoes dehydrobromination where a molecule of hydrobromide gets eliminated from $(E) - 3 - $bromo$ - 3 - $hexene. A dehydrobromination reaction is a kind of elimination reaction where elimination of hydrogen bromide from a substrate takes place. This reaction is mainly associated with the synthesis of alkenes. The reaction of $(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ is given below.

Hence option A is correct that is $(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ gives $3 - $hexyne.
Note : We have approached this reaction with the help of saytzeff rule which favours the alkene with less number of hydrogen on double bonded carbon atoms.As the reaction is dehydrobromination so saytzeff rule is applicable here. We know that during the elimination reaction proton is removed from that carbon atom which have less number of substituents.Therefore $(E) - 3 - $bromo$ - 3 - $hexene when treated with $C{H_3}{O^ - }$ in $C{H_3}OH$ gives $3 - $hexyne.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




