Dacron is an example of:
(a) Polyamides
(b) Polypropenes
(c) Polyacrylonitrile
(d) Polyesters
Answer
262.2k+ views
Hint: Dacron is an example of the family of polymers which is a petroleum-based synthetic fibre. These contain ester groups.
Complete step by step solution:
-Polyester is a category of polymers which contain ester functional groups. Dacron contains an ester functional group.
-Polyethylene terephthalate also known as Dacron is a most common thermoplastic polymer resin of polyester family of polymers. It’s commonly abbreviated as PET or PETE etc.
-IT is used in fibres for clothing, containers for liquids and foods. It is also used in combination with glass fibre for engineering resins.
-Its chemical representation is
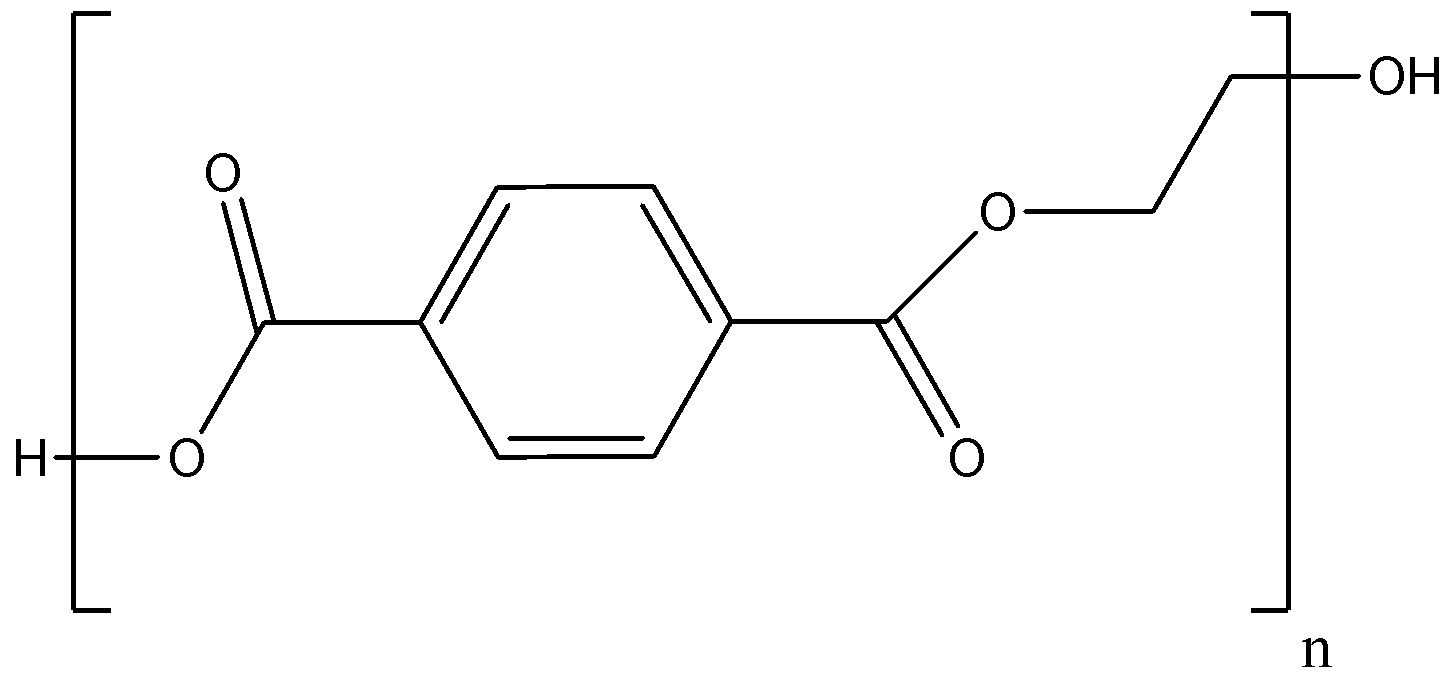
-Its monomer units are ethylene terephthalate. The monomer bis(2-hydroxyethyl) terephthalate is synthesized by the esterification reaction between terephthalic acid and ethylene glycol. It can also be synthesized by transesterification between ethylene glycol and dimethyl terephthalate(DMT) with methanol as a by-product.
-Dacron or PET in its natural state is colourless, semi-crystalline resin. When processed, it becomes semi-rigid and lightweight. It is very strong and is impact-resistant. When exposed to chloroform or other certain chemicals, PET turns white.
-Plastic bottles are made from PET which is used for soft drinks. When modified can be used for flexible food packaging and thermal insulation (such as space blankets). IT has a high mechanical strength and due to this it is also used in tape applications, such as magnetic tape or backing for pressure-sensitive adhesive tapes.
Thus, as Dacron consists of ester groups, it is a polyester and the option (d) is the correct answer to the question.
Note: There are many other names by which Darcon is known. It is known as Terylene in the United Kingdom and it is known as Lavsan in Russia. The name “Dacron” is mainly used in the United States.
Complete step by step solution:
-Polyester is a category of polymers which contain ester functional groups. Dacron contains an ester functional group.
-Polyethylene terephthalate also known as Dacron is a most common thermoplastic polymer resin of polyester family of polymers. It’s commonly abbreviated as PET or PETE etc.
-IT is used in fibres for clothing, containers for liquids and foods. It is also used in combination with glass fibre for engineering resins.
-Its chemical representation is

-Its monomer units are ethylene terephthalate. The monomer bis(2-hydroxyethyl) terephthalate is synthesized by the esterification reaction between terephthalic acid and ethylene glycol. It can also be synthesized by transesterification between ethylene glycol and dimethyl terephthalate(DMT) with methanol as a by-product.
-Dacron or PET in its natural state is colourless, semi-crystalline resin. When processed, it becomes semi-rigid and lightweight. It is very strong and is impact-resistant. When exposed to chloroform or other certain chemicals, PET turns white.
-Plastic bottles are made from PET which is used for soft drinks. When modified can be used for flexible food packaging and thermal insulation (such as space blankets). IT has a high mechanical strength and due to this it is also used in tape applications, such as magnetic tape or backing for pressure-sensitive adhesive tapes.
Thus, as Dacron consists of ester groups, it is a polyester and the option (d) is the correct answer to the question.
Note: There are many other names by which Darcon is known. It is known as Terylene in the United Kingdom and it is known as Lavsan in Russia. The name “Dacron” is mainly used in the United States.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




