Benzyl chloride when oxidised by Lead nitrate solution gives
A. Benzoic acid
B. Benzaldehyde
C. Benzene
D. None
Answer
264k+ views
Hint: Oxidation is the addition of oxygen to a compound.
It involves the loss of electrons increasing the oxidation state of a compound.
Complete Step by Step Solution:
Here, we are given lead nitrate solution, a mild oxidising agent. It is not as powerful as potassium permanganate.
Lead nitrate solution when treated with benzyl chloride acts as an oxidising agent.
Benzyl chloride undergoes oxidation.
A. Benzoic acid
Lead nitrate solution is a mild oxidising agent. It can't convert benzyl chloride directly into benzoic acid.
So, A is incorrect.
B. Lead nitrate solution oxides benzyl chloride into benzaldehyde.
Benzaldehyde does not undergo further oxidation in the presence of a mild oxidizing agent like lead nitrate.
So, B is correct.
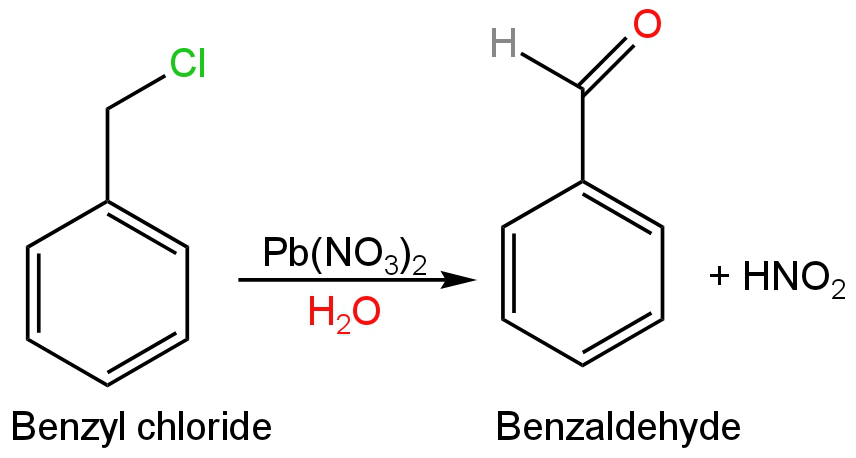
Image: Oxidation of benzyl chloride.
C. Lead nitrate is an oxidising agent that cannot convert benzyl alcohol to benzene.
It is only capable of oxidation.
So, C is incorrect.
So, option B is correct.
Additional Information: Benzyl chloride is a colourless organochlorine compound.
It is prepared by the photochemical reaction of chlorine with benzene.
This reaction follows a free radical mechanism.
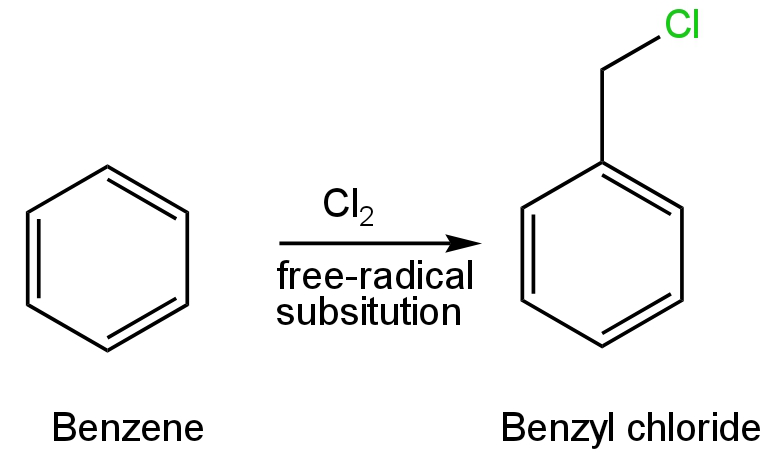
Image: Formation of benzyl chloride
Note: While attempting the question, one must retain in mind that lead nitrate is a mild oxidising agent. When benzyl chloride reacts with alkaline potassium permanganate solution, it undergoes oxidation to form benzoic acid. This happens because of the presence of potassium permanganate which is a powerful oxidising agent. Benzyl chloride in the presence of mild oxidising agents like lead nitrate forms benzaldehyde.
It involves the loss of electrons increasing the oxidation state of a compound.
Complete Step by Step Solution:
Here, we are given lead nitrate solution, a mild oxidising agent. It is not as powerful as potassium permanganate.
Lead nitrate solution when treated with benzyl chloride acts as an oxidising agent.
Benzyl chloride undergoes oxidation.
A. Benzoic acid
Lead nitrate solution is a mild oxidising agent. It can't convert benzyl chloride directly into benzoic acid.
So, A is incorrect.
B. Lead nitrate solution oxides benzyl chloride into benzaldehyde.
Benzaldehyde does not undergo further oxidation in the presence of a mild oxidizing agent like lead nitrate.
So, B is correct.

Image: Oxidation of benzyl chloride.
C. Lead nitrate is an oxidising agent that cannot convert benzyl alcohol to benzene.
It is only capable of oxidation.
So, C is incorrect.
So, option B is correct.
Additional Information: Benzyl chloride is a colourless organochlorine compound.
It is prepared by the photochemical reaction of chlorine with benzene.
This reaction follows a free radical mechanism.

Image: Formation of benzyl chloride
Note: While attempting the question, one must retain in mind that lead nitrate is a mild oxidising agent. When benzyl chloride reacts with alkaline potassium permanganate solution, it undergoes oxidation to form benzoic acid. This happens because of the presence of potassium permanganate which is a powerful oxidising agent. Benzyl chloride in the presence of mild oxidising agents like lead nitrate forms benzaldehyde.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




