Benzamide on reaction with \[POC{l_3}\] gives
(A) Aniline
(B) Chlorobenzene
(C) Benzylamine
(D) Benzonitrile
Answer
265.2k+ views
Hint: The chemical name of \[POC{l_3}\] is phosphoryl chloride. It is used as a dehydrating agent in the given reaction.
Complete Step by Step Solution:
Phosphoryl chloride is a colourless liquid with the formula \[POC{l_3}\]. It hydrolyses in moist air and releases phosphoric acid and fumes of hydrogen chloride. Phosphoryl chloride is a commonly used dehydrating agent in chemical laboratories. It is used as a dehydrating agent for the preparation of nitriles from primary amides.
Benzamide is a white solid with chemical formula \[{C_6}{H_5}CON{H_2}\]. It is the simplest amide derived from benzoic acid. It is soluble in organic solvents and water. These are organic compounds which contain a carboxamide substituent which is attached to a benzene ring. Benzamide is an extremely weak basic compound.
When benzamide reacts with \[POC{l_3}\], a dehydration reaction takes place. The term dehydration means that \[{H_2}O\] is lost from the compound, which results in the formation of the main product that is benzonitrile. Benzonitrile is used as an intermediate for rubber manufacturing, chemicals and as a solvent for nitrile rubber.
The reaction of conversion of benzamide on treatment with \[POC{l_3}\] to give benzonitrile is given below:
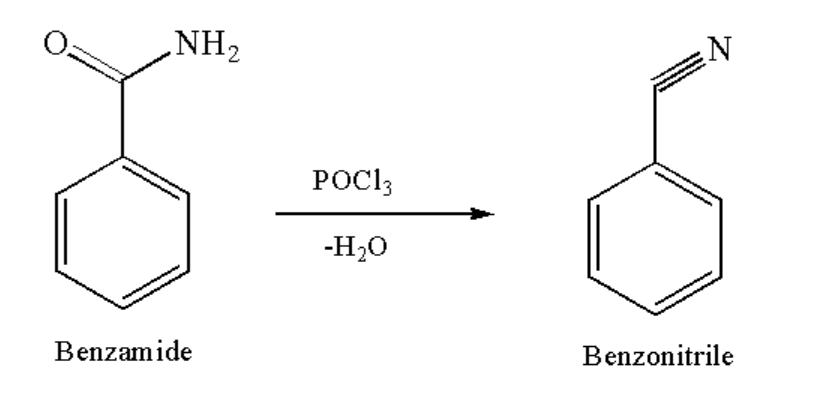
Image: Conversion of benzamide to benzonitrile
So, we can say that Benzonitrile is formed when benzamide reacts with \[POC{l_3}\].
Note: \[POC{l_3}\]is also known as phosphorous oxychloride. Phosphorus oxychloride is synthesised industrially by reacting phosphorus trichloride and oxygen atoms. It can also be prepared by reacting phosphorous trichloride with phosphorus pentachloride. It does not dissolve in water as it forms hydrogen chloride and phosphoric acid by reacting with water.
Complete Step by Step Solution:
Phosphoryl chloride is a colourless liquid with the formula \[POC{l_3}\]. It hydrolyses in moist air and releases phosphoric acid and fumes of hydrogen chloride. Phosphoryl chloride is a commonly used dehydrating agent in chemical laboratories. It is used as a dehydrating agent for the preparation of nitriles from primary amides.
Benzamide is a white solid with chemical formula \[{C_6}{H_5}CON{H_2}\]. It is the simplest amide derived from benzoic acid. It is soluble in organic solvents and water. These are organic compounds which contain a carboxamide substituent which is attached to a benzene ring. Benzamide is an extremely weak basic compound.
When benzamide reacts with \[POC{l_3}\], a dehydration reaction takes place. The term dehydration means that \[{H_2}O\] is lost from the compound, which results in the formation of the main product that is benzonitrile. Benzonitrile is used as an intermediate for rubber manufacturing, chemicals and as a solvent for nitrile rubber.
The reaction of conversion of benzamide on treatment with \[POC{l_3}\] to give benzonitrile is given below:

Image: Conversion of benzamide to benzonitrile
So, we can say that Benzonitrile is formed when benzamide reacts with \[POC{l_3}\].
Note: \[POC{l_3}\]is also known as phosphorous oxychloride. Phosphorus oxychloride is synthesised industrially by reacting phosphorus trichloride and oxygen atoms. It can also be prepared by reacting phosphorous trichloride with phosphorus pentachloride. It does not dissolve in water as it forms hydrogen chloride and phosphoric acid by reacting with water.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




