Aniline and methylamine can be differentiated by
(A) Reaction with chloroform and aqueous solution of KOH
(B) Diazotization followed by coupling with phenol
(C) Reaction with $HN{{O}_{2}}$
(D) None of these
Answer
256.8k+ views
Hint: Both the aniline and methyl amine contain an amino group. The difference is that aniline is an aromatic amine, whereas methylamine is an aliphatic amine. To differentiate these compounds, one of the compounds should give a reaction which the others do not.
Complete Step by Step Solution:
The aniline and methyl amine can be differentiated by the diazotization reaction followed by coupling with phenol. The diazotization reaction is the reaction in which an aromatic amine gets converted into its diazonium salt. Also, the coupling reaction is the reaction in which arene diazonium salt reacts with highly reactive aromatic compounds containing electron-donating groups like$OH$, $N{{H}_{2}}$etc. The diazonium salt of aniline, known as benzene diazonium chloride, is prepared by the reaction of aniline with sodium nitrite $(NaN{{O}_{2}})$ in the presence of an excess of mineral acid. This benzene diazonium chloride will then undergo a coupling reaction at a low temperature (273K-278K) with phenol to form hydroxyazobenzene.
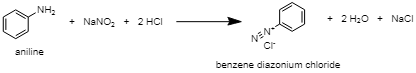
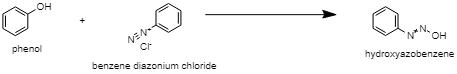
whereas methyl amine does not react with phenols.
Correct Option: (B) Diazotization followed by coupling with phenol.
Additional Information: The benzene diazonium chloride can also react with 2-naphthol to form 2-naphthol aniline dye, which is bright orange in colour. The diazonium salts can be used in the synthesis of various organic compounds. These salts also have applications in nanotechnology and in the dye and pigment industries.
Note: The Benzene diazonium chloride can also react with aniline in the presence of dilute alkali like $NaOH$ in acidic medium to form p-aminoazobenzene, which is also known as yellow dye.
Complete Step by Step Solution:
The aniline and methyl amine can be differentiated by the diazotization reaction followed by coupling with phenol. The diazotization reaction is the reaction in which an aromatic amine gets converted into its diazonium salt. Also, the coupling reaction is the reaction in which arene diazonium salt reacts with highly reactive aromatic compounds containing electron-donating groups like$OH$, $N{{H}_{2}}$etc. The diazonium salt of aniline, known as benzene diazonium chloride, is prepared by the reaction of aniline with sodium nitrite $(NaN{{O}_{2}})$ in the presence of an excess of mineral acid. This benzene diazonium chloride will then undergo a coupling reaction at a low temperature (273K-278K) with phenol to form hydroxyazobenzene.


whereas methyl amine does not react with phenols.
Correct Option: (B) Diazotization followed by coupling with phenol.
Additional Information: The benzene diazonium chloride can also react with 2-naphthol to form 2-naphthol aniline dye, which is bright orange in colour. The diazonium salts can be used in the synthesis of various organic compounds. These salts also have applications in nanotechnology and in the dye and pigment industries.
Note: The Benzene diazonium chloride can also react with aniline in the presence of dilute alkali like $NaOH$ in acidic medium to form p-aminoazobenzene, which is also known as yellow dye.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




