An electron, a proton, a deuteron, and an alpha particle, each having speed are in a region of constant magnetic field perpendicular to the direction of the velocities of the particles. The radius of the circular orbits of these particles is respectively ${{R}_{e,}}{{R}_{p}},{{R}_{d}}$and ${{R}_{\alpha }}$. It follows that
A. ${{R}_{e}}={{R}_{p}}$
B.${{R}_{p}}={{R}_{d}}$
C.${{R}_{d}}={{R}_{\alpha }}$
D.${{R}_{p}}={{R}_{\alpha }}$
Answer
262.8k+ views
Hint: When a charged particle moves with definite velocity and enters a uniform magnetic field $B$, then it experiences a magnetic force perpendicular to the direction of motion and it travels a circular path. Then by equating magnetic force with centripetal force, we can derive the equation of the radius of a circular path.
Formula used:
The radius,$R$ of the circular path in a magnetic field,$B$can be expressed in the following way:
$R=\dfrac{mv}{qB}$
Here $m\And v$are the mass and velocity of the particle with charge $q$.
Complete answer:
When a particle carrying charge$q$, moving with velocity $\vec{v}$enters into a magnetic field $\vec{B}$, it experiences a magnetic force, $\vec{F}=q(\vec{B}\times \vec{v})$
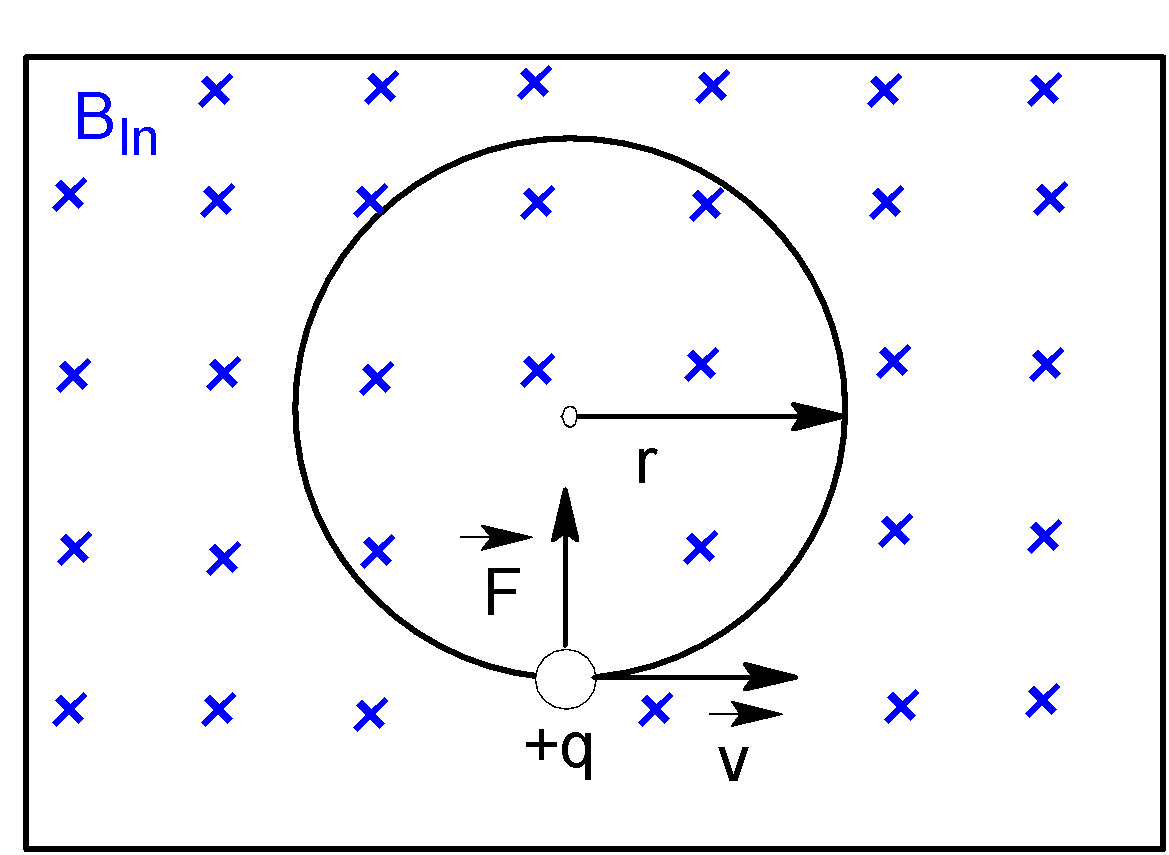
Or,$F=q(Bv\sin {{90}^{o}})$ [Since $\vec{B}$is perpendicular to $\vec{v}$]
Or,$F=qBv$ ……..(i)
As a particle moves in a circular path, then magnetic force becomes a centripetal force $\dfrac{m{{v}^{2}}}{R}$.
Hence by equating magnetic force with centripetal force,
$qBv=\dfrac{m{{v}^{2}}}{R}$
Or,$R=\dfrac{mv}{qB}$
Here we have four charged particles: an electron($e$), a proton ($p$), a deuteron ($d$), and an alpha particle $(\alpha )$. They all have equal speed,$v$and move in a region of the constant magnetic field,$B$.
Therefore the radius of the circular path mainly depends on $\dfrac{mass(m)}{ch\arg e(q)}$ratio.
Or,$R$ $\alpha $ $\dfrac{m}{q}$
Let us check $\dfrac{m}{q}$ratio of each charged particle in the following table,
Let the Mass of a proton be $m$and charge $q$.
As we know the mass of an electron, deuterium and an alpha particle are $\dfrac{1}{1836}$, $2$ and $4$ times the mass of the proton.
Therefore $\dfrac{m}{q}$ ratio for deuterium and an alpha particle are equal, hence their radius of the circular orbit would be equal i.e,${{R}_{d}}={{R}_{\alpha }}$.
Thus, option (C) is correct.
Note:Neutron does not feel any magnetic force while other charged particles experience that force. A neutron is a neutral particle, having no charge. But for charged particle trajectory curvature is proportional to the mass by charge ratio for a definite velocity.
Formula used:
The radius,$R$ of the circular path in a magnetic field,$B$can be expressed in the following way:
$R=\dfrac{mv}{qB}$
Here $m\And v$are the mass and velocity of the particle with charge $q$.
Complete answer:
When a particle carrying charge$q$, moving with velocity $\vec{v}$enters into a magnetic field $\vec{B}$, it experiences a magnetic force, $\vec{F}=q(\vec{B}\times \vec{v})$

Or,$F=q(Bv\sin {{90}^{o}})$ [Since $\vec{B}$is perpendicular to $\vec{v}$]
Or,$F=qBv$ ……..(i)
As a particle moves in a circular path, then magnetic force becomes a centripetal force $\dfrac{m{{v}^{2}}}{R}$.
Hence by equating magnetic force with centripetal force,
$qBv=\dfrac{m{{v}^{2}}}{R}$
Or,$R=\dfrac{mv}{qB}$
Here we have four charged particles: an electron($e$), a proton ($p$), a deuteron ($d$), and an alpha particle $(\alpha )$. They all have equal speed,$v$and move in a region of the constant magnetic field,$B$.
Therefore the radius of the circular path mainly depends on $\dfrac{mass(m)}{ch\arg e(q)}$ratio.
Or,$R$ $\alpha $ $\dfrac{m}{q}$
Let us check $\dfrac{m}{q}$ratio of each charged particle in the following table,
Let the Mass of a proton be $m$and charge $q$.
| Proton | Electron | deuteron | deuteron | |
| Mass($m$) | $m$ | $\dfrac{m}{1836}$ | $2m$ | $4m$ |
| Charge($q$) | $q$ | $q$ | $q$ | $2q$ |
| $\dfrac{mass}{ch\arg e}\left( \dfrac{m}{q} \right)$ | $\dfrac{m}{q}$ | $\dfrac{m}{q\times 1836}$ | $\dfrac{2m}{q}$ | $\dfrac{4m}{2q}=\dfrac{2m}{q}$ |
As we know the mass of an electron, deuterium and an alpha particle are $\dfrac{1}{1836}$, $2$ and $4$ times the mass of the proton.
Therefore $\dfrac{m}{q}$ ratio for deuterium and an alpha particle are equal, hence their radius of the circular orbit would be equal i.e,${{R}_{d}}={{R}_{\alpha }}$.
Thus, option (C) is correct.
Note:Neutron does not feel any magnetic force while other charged particles experience that force. A neutron is a neutral particle, having no charge. But for charged particle trajectory curvature is proportional to the mass by charge ratio for a definite velocity.
Recently Updated Pages
Circuit Switching vs Packet Switching: Key Differences Explained

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
CBSE Class 12 Physics Question Paper 2026: Download SET-wise PDF with Answer Key & Analysis

JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

How to Convert a Galvanometer into an Ammeter or Voltmeter




