An alkene on ozonolysis gives formaldehyde and. The alkene is
A. Ethene
B. Propene
C. 1-butene
D. 2-butene
Answer
257.7k+ views
Hint: Ozonolysis of alkene is the oxidation of alkenes with the help of ozone which gives aldehydes, ketones, or carboxylic acid as the products. This is a redox reaction in organic chemistry one or more than one product can be formed due to this reaction.
Complete Step by Step Answer:
Ozonolysis of alkene goes through the process of oxidative cleavage. Ozonide is formed when ozone attacks the alkene and ozone breaks the π-bond as well as the σ-bond between the carbon and to break the sigma(σ) bond between the carbons high amount of energy and a very reactive species is required and Ozone is a very strong oxidising agent. Ozonolysis of an alkene with no alkyl substituent will produce aldehyde. When one carbon of alkene has both alkyl substituents it will produce ketone and aldehyde and ketones, and further oxidation will produce carboxylic acid.
The give compounds on ozonolysis will produce the following products:
$CH_2=CH_2\ +\ O_3 \longrightarrow \ 2HCHO$
$CH_3CH=CH_2\ +\ O_3 \longrightarrow \ CH_3CHO\ +\ HCHO$
$CH_3CH_2CH=CH_2\ +\ O_3 \longrightarrow \ CH_3CH_2CHO\ +\ HCHO$
$CH_3CH=CHCH_3+ \ O_3 \longrightarrow \ 2CH_3CHO$
As we can see, propene on ozonolysis produces formaldehyde and acetaldehyde as the product.
The reaction of ozonolysis of alkene proceeds through the following mechanism:
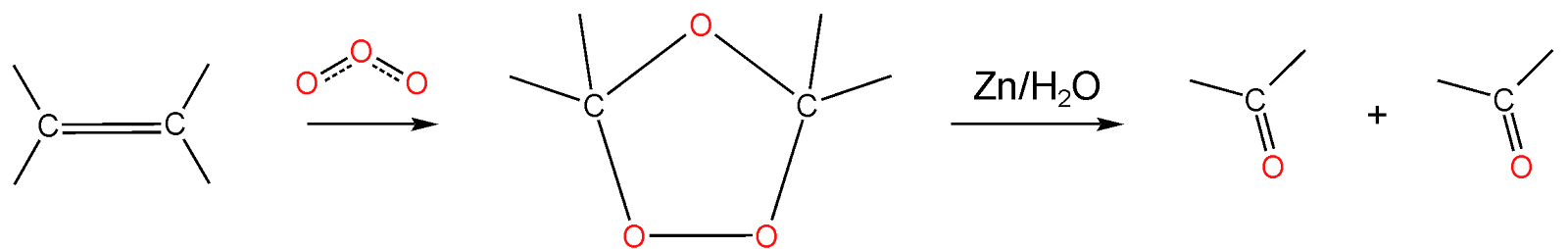
Alkene reacts with the ozone and forms ozonide then the ozonide is further hydrolyzed in presence of Zn to produce aldehydes, ketone, or carboxylic acid. The five-membered heterocyclic ring formed in the reaction is called ozonide.
Thus, Option (B) is correct
Note: Alkenes are also known as olefins. Ozonides are generally formed by alkenes and they are very reactive species; they decompose rapidly into aldehydes, ketone, and peroxide. During the hydrolysis of ozonide hydrogen peroxide (H2O2) is generated to reduce the H2O2. Zinc dust is used as a catalyst in the reaction.
Complete Step by Step Answer:
Ozonolysis of alkene goes through the process of oxidative cleavage. Ozonide is formed when ozone attacks the alkene and ozone breaks the π-bond as well as the σ-bond between the carbon and to break the sigma(σ) bond between the carbons high amount of energy and a very reactive species is required and Ozone is a very strong oxidising agent. Ozonolysis of an alkene with no alkyl substituent will produce aldehyde. When one carbon of alkene has both alkyl substituents it will produce ketone and aldehyde and ketones, and further oxidation will produce carboxylic acid.
The give compounds on ozonolysis will produce the following products:
$CH_2=CH_2\ +\ O_3 \longrightarrow \ 2HCHO$
$CH_3CH=CH_2\ +\ O_3 \longrightarrow \ CH_3CHO\ +\ HCHO$
$CH_3CH_2CH=CH_2\ +\ O_3 \longrightarrow \ CH_3CH_2CHO\ +\ HCHO$
$CH_3CH=CHCH_3+ \ O_3 \longrightarrow \ 2CH_3CHO$
As we can see, propene on ozonolysis produces formaldehyde and acetaldehyde as the product.
The reaction of ozonolysis of alkene proceeds through the following mechanism:

Alkene reacts with the ozone and forms ozonide then the ozonide is further hydrolyzed in presence of Zn to produce aldehydes, ketone, or carboxylic acid. The five-membered heterocyclic ring formed in the reaction is called ozonide.
Thus, Option (B) is correct
Note: Alkenes are also known as olefins. Ozonides are generally formed by alkenes and they are very reactive species; they decompose rapidly into aldehydes, ketone, and peroxide. During the hydrolysis of ozonide hydrogen peroxide (H2O2) is generated to reduce the H2O2. Zinc dust is used as a catalyst in the reaction.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




