An acyl halide is formed when \[PC{l_5}\] reacts with an:
A. Acid
B. Alcohol
C. Amide
D. Ester
Answer
249.9k+ views
Hint: Let us first understand some basic concepts before we proceed with the solution for this question:
Acyl halides can be explained as the halogen substituted hydroxyl groups in organic compounds. Acyl halides are basically formed when the hydroxy or the -OH group of the hydroxyl or -COOH group gets replaced by a halogen. This is a basic substitution reaction.
Complete Step-by-Step Answer:
In order to solve this question, we must find the products formed by \[PC{l_5}\] with all the given options.
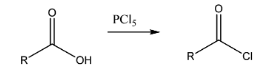
Reaction with acid: the reaction of \[PC{l_5}\] with a given acid would result in the formation of acid chlorides in the products. Acid chloride is the common name for acyl halide where chlorine is the halogen.
Reaction with alcohol: alcohols react in a rather violent manner with \[PC{l_5}\], resulting in the emissive release of hydrochloric acid.
\[R - OH + PC{l_5} \to R - Cl + POC{l_3} + HCl\]
Reaction with amide: when amides are reacted with \[PC{l_5}\], it results in the dehydration of the amide. The corresponding reaction can be given as:
\[R - CON{H_2} + PC{l_5} \to R - C{(Cl)_2}N{H_2} + PC{l_3}\]
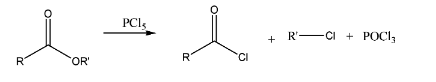
Reaction with esters: reaction between esters and \[PC{l_5}\] results in the formation of an alkyl halide, an acyl halide and also phosphorous oxychloride. Hence, only acyl halide cannot be obtained from esters.
Hence, Option A is the correct option.
Note: In the reaction of \[PC{l_5}\] with carboxylic acid, the reaction goes firstly by a cyclic transition state with the removal of HCl. Then via a nucleophilic addition of chloride where the carbonyl is simultaneously protonated.
Acyl halides can be explained as the halogen substituted hydroxyl groups in organic compounds. Acyl halides are basically formed when the hydroxy or the -OH group of the hydroxyl or -COOH group gets replaced by a halogen. This is a basic substitution reaction.
Complete Step-by-Step Answer:
In order to solve this question, we must find the products formed by \[PC{l_5}\] with all the given options.
Reaction with acid: the reaction of \[PC{l_5}\] with a given acid would result in the formation of acid chlorides in the products. Acid chloride is the common name for acyl halide where chlorine is the halogen.

Reaction with alcohol: alcohols react in a rather violent manner with \[PC{l_5}\], resulting in the emissive release of hydrochloric acid.
\[R - OH + PC{l_5} \to R - Cl + POC{l_3} + HCl\]
Reaction with amide: when amides are reacted with \[PC{l_5}\], it results in the dehydration of the amide. The corresponding reaction can be given as:
\[R - CON{H_2} + PC{l_5} \to R - C{(Cl)_2}N{H_2} + PC{l_3}\]
Reaction with esters: reaction between esters and \[PC{l_5}\] results in the formation of an alkyl halide, an acyl halide and also phosphorous oxychloride. Hence, only acyl halide cannot be obtained from esters.

Hence, Option A is the correct option.
Note: In the reaction of \[PC{l_5}\] with carboxylic acid, the reaction goes firstly by a cyclic transition state with the removal of HCl. Then via a nucleophilic addition of chloride where the carbonyl is simultaneously protonated.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




