Allyl cyanide has:
A.9 sigma bonds and 4 pi bonds
B.9 sigma bonds ,3 pi bonds and 1 lone pair
C.8 sigma bonds and 5 pi bonds
D.8 sigma bonds ,3 pi bonds
Answer
255.3k+ views
Hint: Allyl cyanide means \[C = C\] part attached to cyanide group through a \[C{H_2}\]
group. We have to draw the structure of allyl cyanide and count the number of types of bond present in it. In an easy way we can take it as follows,
1.Single bond contains one sigma bond
2.Double bond contains one sigma and one pi bond
3.Triple bond contains one sigma and two pi bonds.
Number of lone pairs can be counted by looking at the valence shell electrons of all atoms participating in the bond.
Sigma bond \[(\sigma )\] is formed through the overlapping of two orbits along the internuclear axis (the line passing through the center of two nuclei).
pi bond \[(\pi )\] is formed through the overlap between two orbitals perpendicular to the internuclear axis, that is sidewise overlap. According to hybridization concept, hybridized orbital overlap to form sigma bond and unhybridized p orbital forms pi bond.
Complete step by step solution:
The chemical formula of allyl cyanide is \[{C_4}{H_5}N\] .The chemical structure of allyl cyanide is given.

In this structure there are \[C - H\] bonds, \[C - C\] bonds and \[C - N\] bonds. So, we have to check which type of bonds are present in \[C - H\] , \[C - C\] and \[C - N\] bonds. For this we have to know valence shell electron configuration and how this bond formed between atoms.
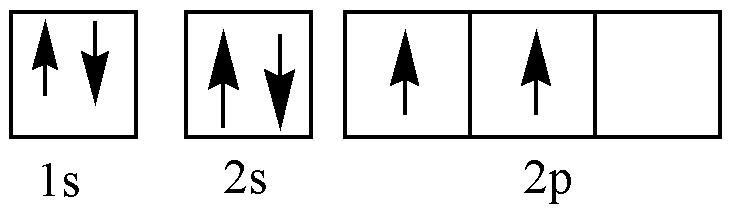
C (ground state):
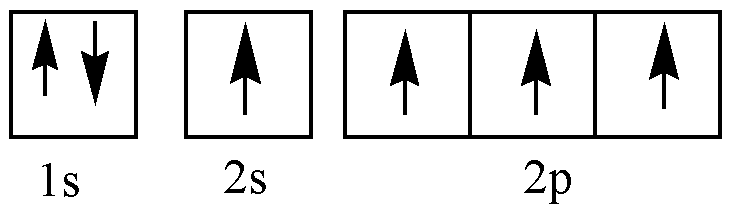
C (excited state):
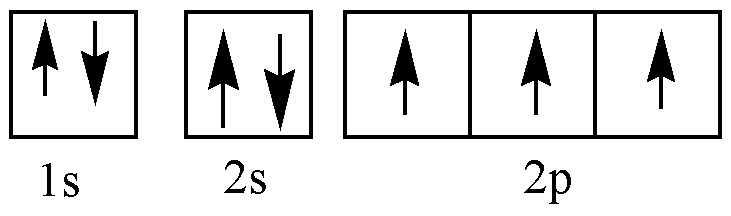
N (ground state):
There are 4 valence electrons in the carbon atom, 5 valence electrons in the nitrogen atom and 1 electron in the hydrogen atom. \[C - H\] bond is formed through the overlap between 2s orbital of carbon and 1s orbital of hydrogen. Overlap between s-s orbital will always be head-on overlap, that is overlap takes place along the internuclear axis. So \[C - H\] bonds are sigma bonds.
\[C - C\] bonds can be formed by the head-on overlap of \[2{p_x}\] orbital of 2 carbon atom and sidewise overlap of \[2{p_y}\] or \[2{p_z}\] orbitals of two carbon atoms. Head-on overlap of p orbital results in sigma bond and sidewise overlap results in pi bonds. That is a double bond contains 1 sigma and 1 pi, the triple bond contains 1 sigma and 2 pi bonds.
In \[C - N\] triple bond two pi bonds and one sigma bond is present. One 2s and one of the 2p orbits will be hybridized. N has 5 valence electrons. Three of them are shared with carbon and the remaining two electrons stay as lone pairs.
Here by looking at the structure we can understand that there are 9 sigma bonds,3 pi bonds and one lone pair of electrons present on the atom. Hence the correct answer is B.
Note: Chemical bond is formed by the force of attraction between atoms, ions or molecules to form a compound. There are different types of chemical bonds, they are ionic, covalent and coordinate bonds. Ionic bond is formed by the transfer of electrons between atoms, covalent bond is formed by sharing of electrons and coordinate bond is a type of bond in which both the shared electrons are provided by one of the atoms. Hydrogen bonding is another type of bonding which is the weakest interaction. It is a type of van der waals interaction. Covalent bond is the strongest bond among these.
group. We have to draw the structure of allyl cyanide and count the number of types of bond present in it. In an easy way we can take it as follows,
1.Single bond contains one sigma bond
2.Double bond contains one sigma and one pi bond
3.Triple bond contains one sigma and two pi bonds.
Number of lone pairs can be counted by looking at the valence shell electrons of all atoms participating in the bond.
Sigma bond \[(\sigma )\] is formed through the overlapping of two orbits along the internuclear axis (the line passing through the center of two nuclei).
pi bond \[(\pi )\] is formed through the overlap between two orbitals perpendicular to the internuclear axis, that is sidewise overlap. According to hybridization concept, hybridized orbital overlap to form sigma bond and unhybridized p orbital forms pi bond.
Complete step by step solution:
The chemical formula of allyl cyanide is \[{C_4}{H_5}N\] .The chemical structure of allyl cyanide is given.

In this structure there are \[C - H\] bonds, \[C - C\] bonds and \[C - N\] bonds. So, we have to check which type of bonds are present in \[C - H\] , \[C - C\] and \[C - N\] bonds. For this we have to know valence shell electron configuration and how this bond formed between atoms.
C (ground state):

C (excited state):

N (ground state):

There are 4 valence electrons in the carbon atom, 5 valence electrons in the nitrogen atom and 1 electron in the hydrogen atom. \[C - H\] bond is formed through the overlap between 2s orbital of carbon and 1s orbital of hydrogen. Overlap between s-s orbital will always be head-on overlap, that is overlap takes place along the internuclear axis. So \[C - H\] bonds are sigma bonds.
\[C - C\] bonds can be formed by the head-on overlap of \[2{p_x}\] orbital of 2 carbon atom and sidewise overlap of \[2{p_y}\] or \[2{p_z}\] orbitals of two carbon atoms. Head-on overlap of p orbital results in sigma bond and sidewise overlap results in pi bonds. That is a double bond contains 1 sigma and 1 pi, the triple bond contains 1 sigma and 2 pi bonds.
In \[C - N\] triple bond two pi bonds and one sigma bond is present. One 2s and one of the 2p orbits will be hybridized. N has 5 valence electrons. Three of them are shared with carbon and the remaining two electrons stay as lone pairs.
Here by looking at the structure we can understand that there are 9 sigma bonds,3 pi bonds and one lone pair of electrons present on the atom. Hence the correct answer is B.
Note: Chemical bond is formed by the force of attraction between atoms, ions or molecules to form a compound. There are different types of chemical bonds, they are ionic, covalent and coordinate bonds. Ionic bond is formed by the transfer of electrons between atoms, covalent bond is formed by sharing of electrons and coordinate bond is a type of bond in which both the shared electrons are provided by one of the atoms. Hydrogen bonding is another type of bonding which is the weakest interaction. It is a type of van der waals interaction. Covalent bond is the strongest bond among these.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




