A sugar solution with sugar molecules and water molecules is-
A. Weak electrolyte
B. Non-electrolyte
C. Metallic conductor
D. Strong electrolyte
Answer
246.3k+ views
Hint: Sugar is sucrose and is composed of twelve carbon atoms, twenty two hydrogen atoms and eleven oxygen atoms which are bonded together by covalent bonds. Water is a polar molecule composed of two hydrogen atoms and one oxygen atom which are also bonded together by covalent bond.
Step-by-Step Solution-
Sugar solution contains both sugar molecules and water molecules. Sugar molecules have a chemical formula ${C_{12}}{H_{22}}{O_{11}}$ where all the carbon, hydrogen and oxygen atoms are joined together by covalent bonds. Its structure is-
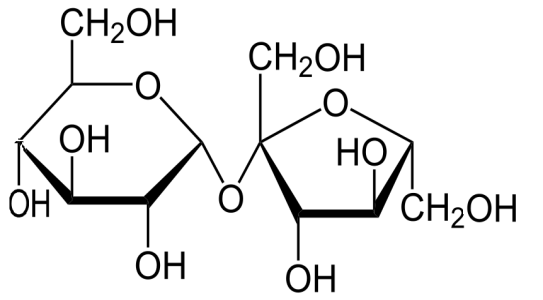
Since it is covalently bonded so it cannot decompose into ions which are necessary to conduct electricity in the solution.
Water molecule has formula ${{\text{H}}_2}{\text{O}}$ and it does dissociate into ions but its ionization constant is very low so the numbers of ions produced are not enough to conduct electricity. Since sugar and water molecules do not readily dissociate into ions when they are in their dissolved state and do not conduct electricity. Hence they are Non-electrolytes.
Non- electrolytes are typically joined by covalent bonds unlike electrolytes which are held together by ionic bonds so non- electrolytes do not easily break apart when placed in the aqueous solution.
Hence the correct answer is B.
Note: Electrolytes are totally opposite of non-electrolytes as they conduct electricity when they are in their dissolved state as they readily decompose into their ions. There are two types of electrolyte –
Weak Electrolyte- They do not dissociate completely in water only partially ionize in aqueous solution like hydrogen fluoride HF.
Strong Electrolyte-They completely dissociate into ions in the aqueous solution and completely ionize like sodium hydroxide (NaOH).
Metallic conductor is a metal that conducts electricity due to flow of electrons like in copper, silver. There is only a physical change involved in it, no chemical changes occur.
Step-by-Step Solution-
Sugar solution contains both sugar molecules and water molecules. Sugar molecules have a chemical formula ${C_{12}}{H_{22}}{O_{11}}$ where all the carbon, hydrogen and oxygen atoms are joined together by covalent bonds. Its structure is-

Since it is covalently bonded so it cannot decompose into ions which are necessary to conduct electricity in the solution.
Water molecule has formula ${{\text{H}}_2}{\text{O}}$ and it does dissociate into ions but its ionization constant is very low so the numbers of ions produced are not enough to conduct electricity. Since sugar and water molecules do not readily dissociate into ions when they are in their dissolved state and do not conduct electricity. Hence they are Non-electrolytes.
Non- electrolytes are typically joined by covalent bonds unlike electrolytes which are held together by ionic bonds so non- electrolytes do not easily break apart when placed in the aqueous solution.
Hence the correct answer is B.
Note: Electrolytes are totally opposite of non-electrolytes as they conduct electricity when they are in their dissolved state as they readily decompose into their ions. There are two types of electrolyte –
Weak Electrolyte- They do not dissociate completely in water only partially ionize in aqueous solution like hydrogen fluoride HF.
Strong Electrolyte-They completely dissociate into ions in the aqueous solution and completely ionize like sodium hydroxide (NaOH).
Metallic conductor is a metal that conducts electricity due to flow of electrons like in copper, silver. There is only a physical change involved in it, no chemical changes occur.
Recently Updated Pages
Which of the following contains the highest percentage class 12 chemistry JEE_Main

Protein can be most easily removed from A Alkanes B class 12 chemistry JEE_Main

PhCOCHBr2xrightarrowOHAxrightarrowOHBxrightarrowH+C class 12 chemistry JEE_Main

When alcohol reacts with conc H2SO4 intermediate compound class 12 chemistry JEE_Main

A nanopeptide contains peptide linkages A 10 B 8 C class 12 chemistry JEE_Main

Which one of the following is paramagnetic A NO B N2O class 12 chemistry JEE_Main

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Collisions: Types and Examples for Students

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The D And F Block Elements - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




