2, 2- Dimethyloxirane can be cleaved by:
(A) Acid
(B) Water
(C) Bromine
(D) None of these
Answer
267.3k+ views
Hint: The structure of 2, 2-dimethyloxirane contains a cyclopropane ring. 2, 2-Dimethyloxirane contains a heterocyclic ring in its structure. 2, 2-Dimethyloxirane also called as 1, 2-isobutylene oxide. The structure of 2, 2- Dimethyloxirane is as follows.
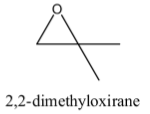
Complete step by step solution:-
-The ring strain is too high in cyclopropane rings when compared to cyclobutane, cyclopentane and cyclohexane.
-Because of ring strain in the structure of 2, 2-dimethyloxirane, it will undergo cleavage very easily.
-The cleavage of the 2, 2-dimethyloxirane is as follows.
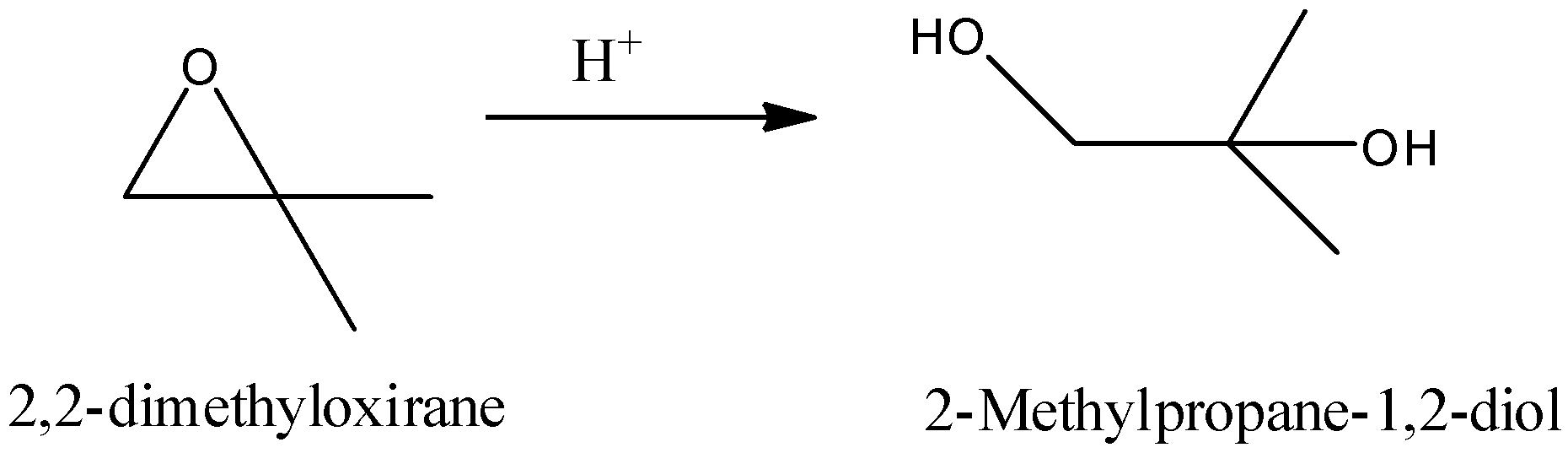
-The mechanism of the cleavage of the 2, 2-dimethyloxirane is as follows.
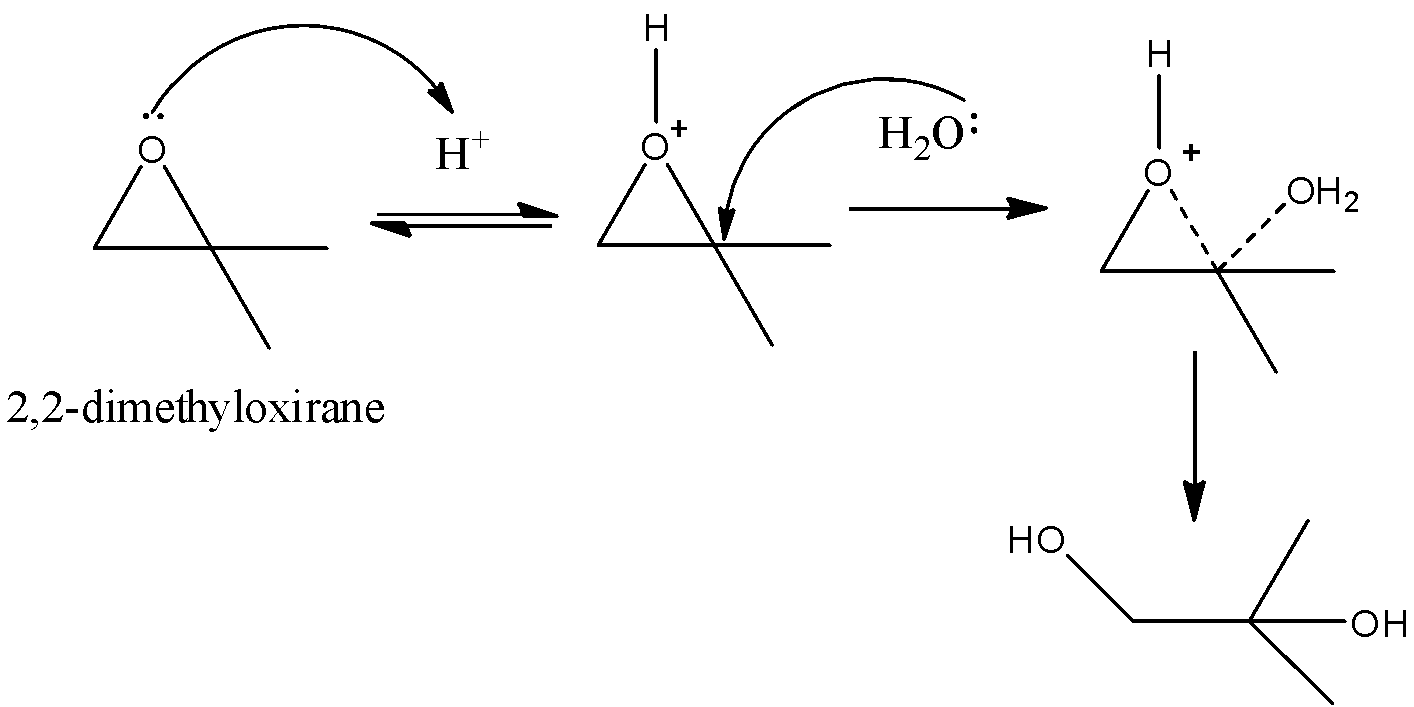
-In the above mechanism we can see clearly that the ring cleavage is going to be initiated by an acid.
-In the first step the acid is going to form a bond with oxygen which is present in the ring and oxygen gets a positive charge because oxygen forms three bonds with the acid.
-Later the lone pair of electrons in the water molecule reacts with tertiary carbon atoms in the ring.
-Then the ring slowly destabilizes and the ring is going to cleave and forms an acyclic compound.
-Therefore the oxirane ring in 2, 2- dimethyloxirane is cleaved by the presence of an acid.
So, the correct option is A.
Note: Because of the presence of ring strain in the given molecule 2,2- dimethyloxirane, it undergoes ring cleavage easily with very small amounts of the acid. Cyclopropane has the highest strain in the ring of 114kJ/mol.
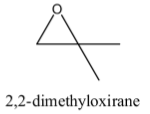
Complete step by step solution:-
-The ring strain is too high in cyclopropane rings when compared to cyclobutane, cyclopentane and cyclohexane.
-Because of ring strain in the structure of 2, 2-dimethyloxirane, it will undergo cleavage very easily.
-The cleavage of the 2, 2-dimethyloxirane is as follows.

-The mechanism of the cleavage of the 2, 2-dimethyloxirane is as follows.

-In the above mechanism we can see clearly that the ring cleavage is going to be initiated by an acid.
-In the first step the acid is going to form a bond with oxygen which is present in the ring and oxygen gets a positive charge because oxygen forms three bonds with the acid.
-Later the lone pair of electrons in the water molecule reacts with tertiary carbon atoms in the ring.
-Then the ring slowly destabilizes and the ring is going to cleave and forms an acyclic compound.
-Therefore the oxirane ring in 2, 2- dimethyloxirane is cleaved by the presence of an acid.
So, the correct option is A.
Note: Because of the presence of ring strain in the given molecule 2,2- dimethyloxirane, it undergoes ring cleavage easily with very small amounts of the acid. Cyclopropane has the highest strain in the ring of 114kJ/mol.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




