



Introduction to Carbohydrates - What are Carbohydrates?
Carbohydrates have been defined historically as chemicals with the empirical formula Cn(H2O)n. However, carbohydrate is presently defined as a polyhydroxy aldehyde or polyhydroxy ketone with the conventional formula, a molecule nearly comparable to it, or oligomers or polymers of such molecules. They need a different set of skills to manage than classic "natural products" like terpenes, steroids, and alkaloids since they are water-soluble and difficult to crystallise.
Carbohydrates in chemistry aim to teach the students about the various concepts. This article provides an example of the types of questions that might be asked about this subject in the JEE exam.
Important Topics of Carbohydrates
Structure and Classification of Carbohydrates
Carbohydrates and its Types
Importance of Carbohydrates
Some Important Definitions
Important Terms | Explanation |
Monosaccharides | Monosaccharides are polyhydroxy aldehydes or -ketones with an unbranched C-chain in most cases. All the monosaccharides have the formula as (CH2O) n and n > 3. |
Polysaccharides | They are one of the most common types of biomolecules. They're lengthy carbohydrate molecules made up of numerous smaller monosaccharides. |
Glucose | With six carbon atoms and one aldehyde group, glucose is a simple sugar. The molecular formula for this monosaccharide is C6H12O6. |
Definition of Carbohydrates
Carbs is also known as saccharides, a Greek term that means "sugar," because nearly all carbohydrates have a sweet flavour.
The word carbohydrate or carbon hydrate comes from its fundamental constituent formula, which has carbon linked to hydrogen and oxygen in the same ratio as water. Carbohydrates are polyhydroxy aldehydes or ketones, their simple derivatives, or polymers from a chemical standpoint.
Carbohydrates are "optically active polyhydroxy aldehydes or ketones, or the substances that yield units of such kind on hydrolysis," according to chemistry.
General Formula of Carbohydrates
Carbohydrates have the general formula Cx(H2O)y.
Because carbohydrates (or sugars) contain the general formula Cx(H2O)y, they were once thought to be "carbon hydrates."
They have the same proportions of hydrogen and oxygen as water. It should be noted that certain carbohydrates, such as 2-deoxyribose C5H10O4, do not comply with the formula Cx(H2O)y. The majority of them, however, follow the formula Cx(H2O)y.
Carbohydrates are also known as sugars in general, and there are several partly methylated sugars, amino sugars, and amino sugars that occur naturally, as well as one natural nitro sugar. All carbohydrates are polyhydroxy aldehydes or ketones, or compounds that hydrolyze to produce them.
Sources of Carbohydrates
The following are the sources to obtain carbohydrates:
Bread
Beans
Milk
Popcorn
Potatoes
Cookies
Spaghetti
Soft drinks
Corns
Types of Carbohydrates
Carbohydrates are available in a variety of forms. Generally, carbohydrates are divided into two categories: simple and complicated. This classification is based mostly on their chemical structure and degree of polymerisation.
Simple Carbohydrates: They include one or two sugar molecules. Carbohydrates like these can be found in abundance in dairy products, refined sugar, and other foods. These carbs are considered empty calories since they include no fibre, vitamins, or minerals.
Simple carbs are further broken down into three types.
Monosaccharides: They are carbohydrate molecules that contain only one sugar unit.
Disaccharides: They are formed when two monosaccharides mix. This carbohydrate is found in a variety of forms, including sucrose, lactose, and maltose.
Oligosaccharides: They are carbohydrate molecules that are made up of two to nine monomers.
Complex Carbohydrates: Two or more sugar molecules make up complex carbohydrates. Carbohydrates like these may be found in abundance in foods like corn, lentils, peanuts, beans, and so on. Polymerisation produces complex carbohydrates, which are also known as polysaccharides.
Importance of Carbohydrates (Uses of Carbohydrates)
We come across and consume a lot of carbs in our daily lives. They are an important element of our diet since they provide us with the energy we need to accomplish our jobs. Carbohydrates or saccharides are split into four sub-categories based on the number of monomers present in their structure: monosaccharide, disaccharide, oligosaccharide, and polysaccharide.
Glucose, for example, is a monosaccharide with only one glucose unit. Sucrose is a disaccharide that consists of units of glucose and fructose.
Carbohydrates serve the following purposes:
They supply energy to our bodies. All of the carbohydrates in meals are digested and transformed into a single glucose molecule. Glucose is a monosaccharide that is involved in cellular respiration and creates ATPs, which are the cells' energy currency.
Carbohydrates keep the body's blood sugar levels in check. Insulin is a hormone that controls the quantity of glucose in the blood.
Sweeteners are carbohydrates. The sugar we use in our homes is sucrose, which is a carbohydrate.
Dietary fibres include carbohydrates such as cellulose, pectin, and mucilage, among others.
Carbohydrates are found on cell surfaces and in antibodies, both of which play a role in cell identification.
Carbohydrates serve a variety of other purposes.
Sources of Carbohydrates
Many fruits contain simple sugars in the form of fructose.
All dairy products include galactose.
Lactose may be found in large quantities in milk and other dairy products.
Maltose may be found in a variety of foods, including cereal, beer, potatoes, processed cheese, and pasta.
Sucrose is derived from sugar and honey, both of which include trace quantities of vitamins and minerals.
Milk, fruits, and vegetables all include simple sugars that contain minerals and vitamins.
Many refined and other processed goods, such as white flour, white rice, and sugar, are labelled "enriched" because they lack critical nutrients. Using vitamins, carbs, and all other organic substances in their natural forms is highly healthful.
Examples of Carbohydrates
Some of the common carbohydrates examples are:
Galactose
Maltose
Lactose
Glucose
Fructose
Sucrose
Cellulose
Starch
Structure of Carbohydrates – Glucose
One of the most significant monosaccharides is glucose. The following are the two most frequent ways of preparing glucose.
Sucrose is converted to glucose and fructose when it is cooked with dilute acid in an alcohol solution.
From Starch: Glucose may also be made by hydrolyzing starch and boiling it with weak sulphuric acid at 393 degrees Fahrenheit under high pressure.
Glucose, commonly known as dextrose and aldohexose, is a sugar that is abundant on the planet.
Its structure can be given as
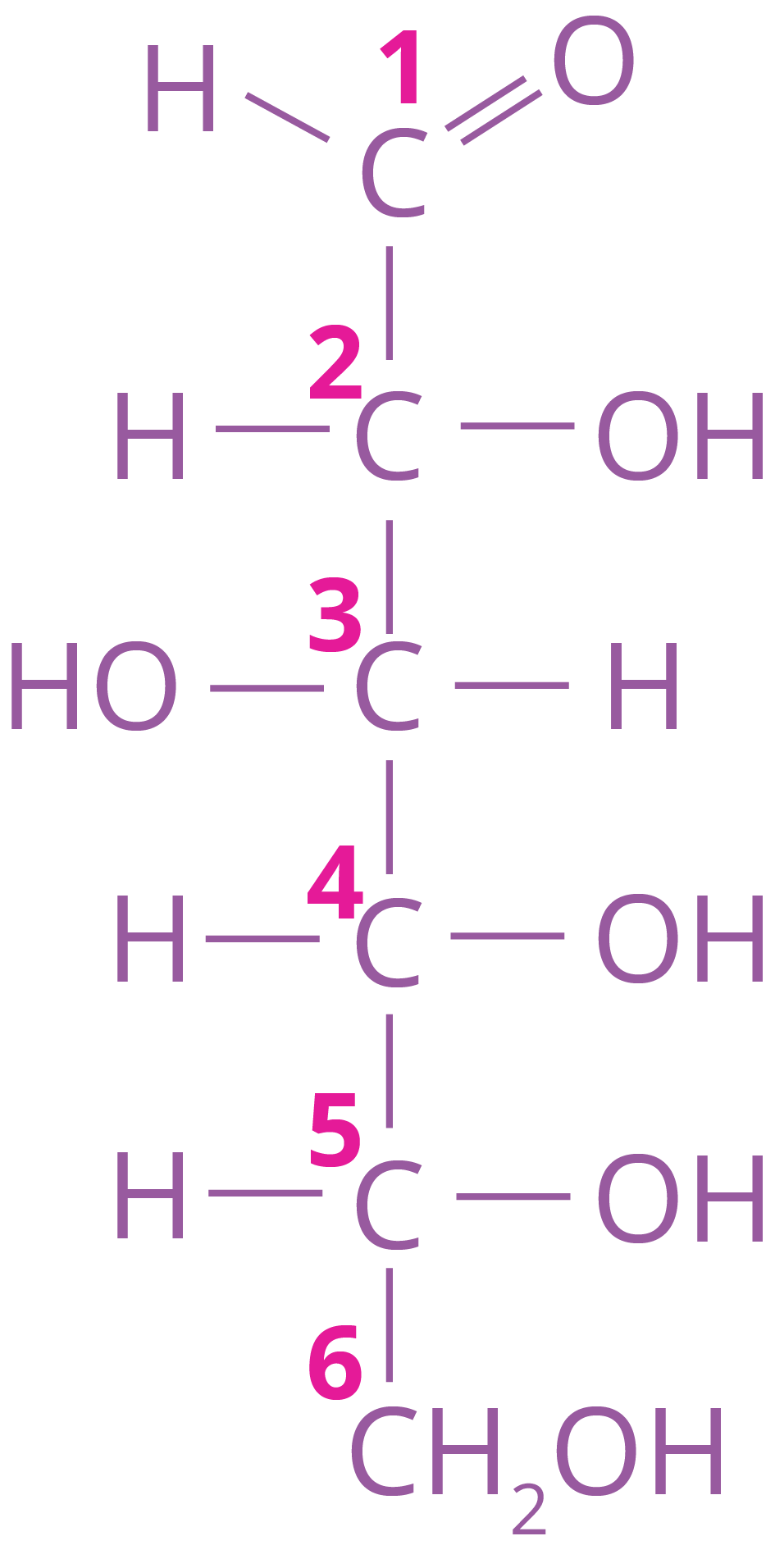
Structure of Carbohydrates – Fructose
Fructose has the chemical formula C6H12O6 and is an essential ketohexose. It includes six carbon atoms in the shape of a straight chain and has a ketone functional group at carbon number 2. Furanose is the name given to the ring component of fructose, which is comparable to the chemical Furan. Fructose has the following structure:
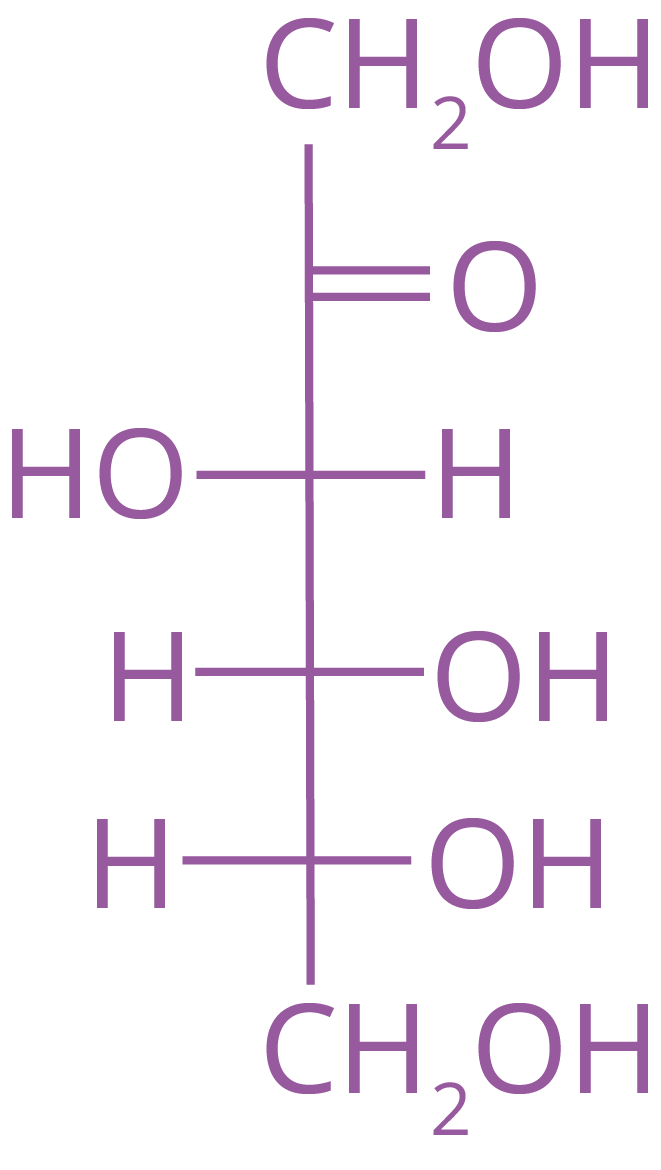
Solved Examples From the Chapter
Example 1: Which of the following monosaccharide is a pentose
(A) Galactose
(B) Glucose
(C) Fructose
(D) Arabinose
Solution:
A pentose is found to be a monosaccharide i.e., simple sugar with five carbon atoms. Option (D) is correct because Arabinose is a pentose monosaccharide while glucose, fructose, and galactose are hexose sugar. Its structure can be given as
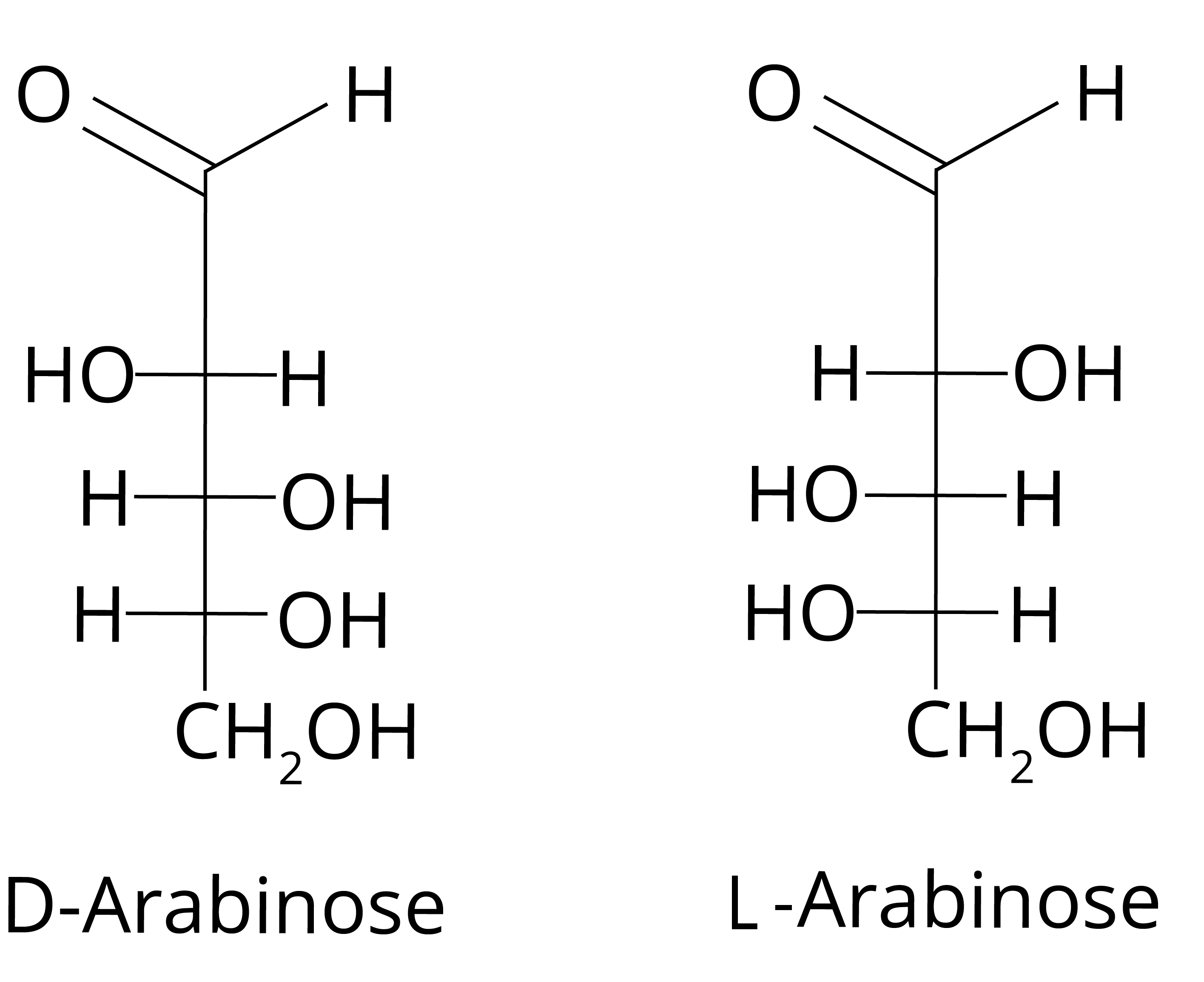
Key Point to Remember: The definition of Pentose sugar and the structures of the given sugars.
Example 2: Guncotton is
A) Nitrosucrose
B) Nitrocellulose
C) Nitroglucose
D) Nitropicrin
Solution:
Guncotton is a nitrocellulose or cellulose trinitrate which is used in explosive and as a binder for solid rocket propellants. Hence option B) is correct.
Key Point to Remember: Guncotton also known as nitrocellulose or trinitrocellulose or cellulose nitrate and is a mild explosive due to which it is used in rockets.
Solved Questions from the Previous Years’ Question Papers
Question 1: General formula for carbohydrates is
(A) CnH2nO2n+2
(B) Cx(H2O)2x
(C) Cx(H2O)y
(D) None of these
Solution:
Carbohydrates have the general formula Cx(H2O)y.
Because carbohydrates (or sugars) contain the general formula Cx(H2O)y, they were once thought to be "carbon hydrates."
So, the correct option is (C).
Trick: Familiarity with the general formula for carbohydrates.
Question 2: The term anomers of glucose refer to
(A) isomers of glucose that differ in configurations at carbons one and four(C-1 and C-4)
(B) a mixture of (D)-glucose and (L)-glucose
(C) enantiomers of glucose
(D) isomers of glucose that differ in configuration at carbon one (C-1)
Solution:
In a cyclic saccharide, an anomer is an epimer at the hemiacetal or hemiketal carbon, an atom known as the anomeric carbon. A stereoisomer is something like an anomer. The carbon atom in position one is an anomeric carbon atom.
As a result, option (D) is the correct answer.
Trick: Anomers are cyclic monosaccharides, differing from each other in the configuration of C-1 carbon or C-2 carbon.
Question 3: Which of the following compounds can be detected by Molisch’s test?
(A) Nitro compounds
(B) Sugars
(C) Amines
(D) Primary alcohols
Solution:
Molisch's test detects the presence of carbohydrates in a sample. The presence of carbohydrates in the analyte is confirmed by the creation of a purple or purplish-red ring at the site of contact between the H2SO4 and the analyte + Molisch's reagent combination. Sugars and starches are the two types of carbohydrates. Molisch's test can therefore identify sugars.
As a result, option (B) is the correct answer.
Trick: Molisch's test is a chemical test which is used to check for the presence of carbohydrates in a given analyte.
Practice Questions
Question 1: The change in optical rotation, with time, of a freshly prepared solution of sugar, is known as
(A) Rotatory motion
(B) Inversion
(C) Specific rotation
(D) Mutarotation
Answer: D) Mutarotation
Question 2: α-D (+)-glucose and β-D(+)-glucose are
(A) conformers
(B) epimers
(C) anomers
(D) enantiomers
Answer: (C) anomers
Conclusion
Glucose is the primary source of energy for human cells. Carbohydrates are responsible for supplying glucose to human body cells, allowing them to perform their essential duties. Carbohydrates also aid in the preservation of muscles and the storage of energy for later usage.
A variety of concepts are involved to study this topic and further help in understanding a phenomenon in detail. Hence, this is important not only for competitive exams like JEE or NEET but also for a better understanding of this topic.
FAQs on JEE Chapter - Carbohydrates
1. What are carbohydrates?
Carbohydrates are sugars, starches, and fibres found in fruits, cereals, vegetables, and milk products. Carbohydrates are the body's principal source of energy, according to the American Diabetes Association. Carbohydrates are named for the chemical elements carbon, hydrogen, and oxygen that they contain.
2. What are the major functions of carbohydrates?
Carbohydrates function includes four main activities in the body: they supply energy, store energy, produce macromolecules, and free up protein and fat for other purposes. The majority of glucose energy is converted to glycogen in the muscle and liver.
3. What are the sources of carbohydrates?
Fresh fruits, tomatoes, maize, potatoes, meat, and milk products are all good sources of carbohydrates, both animal and plant-based. Soda, white bread, added sugar, pastries, and other highly processed foods are examples of foods that are not safe.























