The equation for Freundlich adsorption isotherm is:
A. \[\dfrac{x}{m} = K{p^{1/n}}\]
B. \[x = mK{p^{1/n}}\]
C. \[\dfrac{x}{m} = K{p^{ - n}}\]
D. All of these
Answer
249.6k+ views
Hint: From the name itself, it becomes clear that Freundlich adsorption isotherm is a curve that expresses the variation in the amount of gas adsorbed by the adsorbent with the temperature at the constant pressure.
Complete step-by-step answer:
The Freundlich adsorption isotherm is an empirical relation between the concentrations of a solute on the surface of an adsorbent to the concentration of the solute present in the liquid. Freundlich equation is an expression which represents the isothermal variation of adsorption of a gas adsorbed on a unit mass solid adsorbent with pressure. Adsorption is an attractive interaction between atoms, molecules or ions of a gas or liquid to a surface or deposition of molecular species on a surface called adsorbent.
Its mathematical expression can be analysed as:
\[\dfrac{x}{m} = K{p^{1/n}}\](\[n > {\text{ }}1\])
Where, x is the mass of the adsorbent, k and n are constants that depend on the nature of adsorbent and the gas at a specific temperature .Adsorption factors consists of temperature, pore-volume, degree of saturation, molecular sieve, type of adsorbent, and surface area.
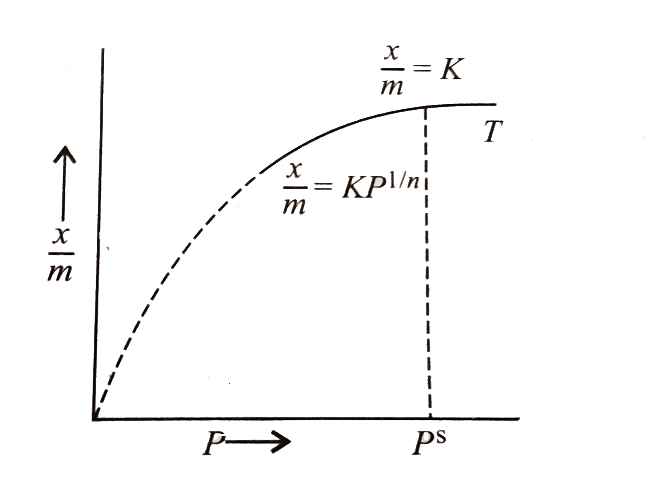
Hence, the correct option is (A).
Note: One might get confused between adsorption and absorption. They seem similar but are totally different as absorption is a bulk process in which the particles go into the bulk of the solvent while adsorption is a surface phenomenon in which the particles remain stuck to the surface of the solvent.
Complete step-by-step answer:
The Freundlich adsorption isotherm is an empirical relation between the concentrations of a solute on the surface of an adsorbent to the concentration of the solute present in the liquid. Freundlich equation is an expression which represents the isothermal variation of adsorption of a gas adsorbed on a unit mass solid adsorbent with pressure. Adsorption is an attractive interaction between atoms, molecules or ions of a gas or liquid to a surface or deposition of molecular species on a surface called adsorbent.
Its mathematical expression can be analysed as:
\[\dfrac{x}{m} = K{p^{1/n}}\](\[n > {\text{ }}1\])
Where, x is the mass of the adsorbent, k and n are constants that depend on the nature of adsorbent and the gas at a specific temperature .Adsorption factors consists of temperature, pore-volume, degree of saturation, molecular sieve, type of adsorbent, and surface area.

Hence, the correct option is (A).
Note: One might get confused between adsorption and absorption. They seem similar but are totally different as absorption is a bulk process in which the particles go into the bulk of the solvent while adsorption is a surface phenomenon in which the particles remain stuck to the surface of the solvent.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




